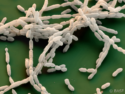
Alginate hydrogels are of great interest for applications such as food, biomedicines, and pharmaceuticals because of their excellent biocompatibility, biodegradability, well-developed derivatization techniques, and variety of natural sources. Direct mixing of cations and alginate leads to inhomogeneous gels due to rapid and irreversible binding. Therefore, aerogels are produced via multiple isolation steps including gelation, solvent exchange, and drying under supercritical conditions.
Pavel Gurikov and Irina Smirnova, Hamburg University of Technology, Germany, Marc Fricke, BASF Polyurethanes GmbH, Lemförde, Germany, and colleagues have developed a one-pot process for the preparation of alginate aerogels that uses high-pressure CO2. Pressurized CO2 at 5 MPa and 298 K acts as a weak acid. This leads CaCO3 suspended in the sodium alginate solution to release calcium ions. The calcium ions crosslink the alginate.
This results in the formation of a hydrogel. Solvent exchange to ethanol at ambient conditions followed by supercritical drying with carbon dioxide results in alginate aerogels with remarkable properties compared to the state of the art.
The hydrogels are very light with densities down to 0.06 – 0.02 g cm–3, translucent and possess a fibrillar structure with both meso- and macroporosity. For selected samples, the surface area and mesopore volume are 545 ± 77 m2 g–1 and 6.98 cm3 g–1, respectively. Thermal conductivity measurements revealed excellent thermal insulation properties.
- A novel approach to alginate aerogels: carbon dioxide induced gelation,
P. Gurikov, S. P. Raman, D. Weinrich, M. Fricke, I. Smirnova,
RSC Adv. 2014, 5, 7812–7818.
DOI: 10.1039/c4ra14653k
Also of Interest
- Insulation Through an Organic Polyurethane-Based Aerogel (Interview with Marc Fricke),
Vera Koester,
ChemViews Mag. 2014.
DOI: 10.1002/chemv.201400093