Coaxing several highly electronegative flourine atoms into close proximity is a challenge for synthesis, but can yield molecules with interesting electrostatic properties.
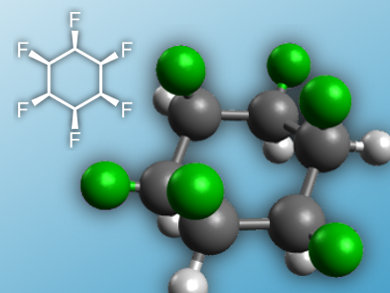
David O’Hagan, University of Saint Andrews, UK, and colleagues have prepared the highest energy configuration of 1,2,3,4,5,6-hexaflourocyclohexane, where all flourine atoms are on the same face of the ring. The team started from myo-inositol, which features six C–OH groups, and converted it to a diepoxidediol with all six oxygen atoms on the same side of the ring in a six-step process. The oxygen substituents of this precusor were then substituted by flourine in another six steps, inverting the configuration at each carbon to yield the desired product.
The researchers characterized the all-cis product by single-crystal X-ray analysis and 19F NMR spectroscopy. The cyclohexane adopts a chair conformation (pictured). Calculations give a value of 6.2 D for the dipole moment, the highest value for a non-ionic aliphatic compound. In the solid state, the molecules are ordered according to the electrostatic interactions. The ring could thus be an interesting building block for supramolecular chemistry.
- All-cis 1,2,3,4,5,6-hexafluorocyclohexane is a facially polarized cyclohexane,
Neil S. Keddie, Alexandra M. Z. Slawin, Tomas Lebl, Douglas Philp, David O’Hagan,
Nat. Chem. 2015.
DOI: 10.1038/nchem.2232