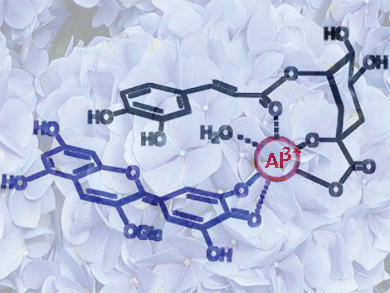
Naturally occurring blue plant colorings, especially the anthocyanins, are notoriously difficult to use as commercial food colorings because of their instability. The natural coloring agent hydrangea blue is an Al3+ complex with varying ratios of the anthocyanin delphinidin 3-O-glucoside and co-pigments 5-O-caffeoylquinic acid and 5-O-p-coumaroylquinic acid. This complex exists in aqueous solution at approximately pH 4.0. Attempts to crystallize it have failed so far.
Kumi Yoshida, Nagoya University, Japan, and colleagues conducted an extensive visible light absorption, circular dichroism, and NMR study of various mixtures of the hydrangea blue component compounds to try to reconstruct this color synthetically and determine the structure of the metal complex. They found that the molar ratios of the components and the structure of the hydrangea blue complex are not fixed, but fluctuate in an equilibrium state in solution. They observed that complexation with Al3+ forms the blue anydrobase anion form of delphinidin 3-O-glucoside, and this complex is stabilized by 5-O-caffeoylquinic acid, which also chelates to Al3+.
- Metal Complex Pigment Involved in the Blue Sepal Color Development of Hydrangea,
Kin-ichi Oyama, Tomomi Yamada, Daisuke Ito, Tadao Kondo, Kumi Yoshida,
J. Agric. Food Chem. 2015.
DOI: 10.1021/acs.jafc.5b02368