[2π+2π] Cycloadditions give access to cyclobutanes. Metal-catalyzed reactions offer a direct and versatile approach for this, but are not without issues. They often suffer from low selectivity and sometimes require functionalized alkenes as starting materials. Additionally, some transition-metal complexes used as catalysts can be highly air- and moisture-sensitive.
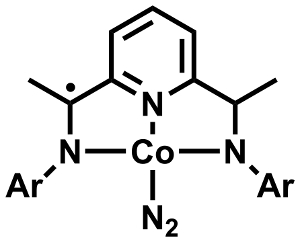 Paul J. Chirik and colleagues, Princeton University, NJ, USA, have developed an aryl-substituted bis(imino)pyridine cobalt catalyst (pictured) in an attempt to solve these problems. Cobalt is less reactive than some of the other transition metals, which makes the catalyst less sensitive to oxygen and water. The team characterized the (paramagnetic) catalyst using electron paramagnetic resonance (EPR) and x-ray crystallography, and used quantum chemical calculations as well as kinetic studies to elucidate the reaction mechanism.
Paul J. Chirik and colleagues, Princeton University, NJ, USA, have developed an aryl-substituted bis(imino)pyridine cobalt catalyst (pictured) in an attempt to solve these problems. Cobalt is less reactive than some of the other transition metals, which makes the catalyst less sensitive to oxygen and water. The team characterized the (paramagnetic) catalyst using electron paramagnetic resonance (EPR) and x-ray crystallography, and used quantum chemical calculations as well as kinetic studies to elucidate the reaction mechanism.
The ligand is redox-active and plays an important part in the reaction. The catalyst first coordinates one alkene moiety and loses its N2 ligand. Upon coordination of the second alkene, the coodination at cobalt switches from planar to tetrahedral and the unpaired electron moves from the ligand to the metal center, where the formed radical promotes the cycloaddition. The product is then released in a final reductive elimination, which the team considers the rate-limiting step.
The catalyst allows the formation of cyclobutanes under mild conditions from unactivated alkenes, and tolerates functional groups such as amines and ethers. The reserchers hope to increase its performance based on their understanding of the reaction mechanism.
- Cobalt-Catalyzed [2π+2π] Cycloadditions of Alkenes: Scope, Mechanism and Elucidation of Electronic Structure of Catalytic Intermediates,
Valerie A. Schmidt, Jordan M Hoyt, Grant W. Margulieux, Paul J. Chirik,
J. Am. Chem. Soc. 2015.
DOI: 10.1021/jacs.5b04034
![Cobalt-Catalyzed [2+2] Cycloadditions](https://www.chemistryviews.org/wp-content/uploads/legacy/common/images/thumbnails/source/14ddd81befa.jpg)



