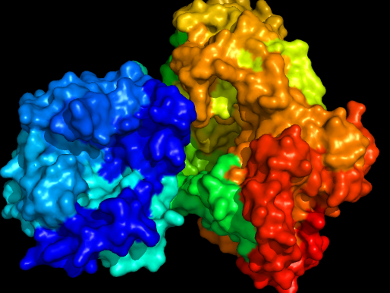
The mosquito-borne dengue virus infects hundreds of millions of people each year, tens of thousands of which die of dengue fever. There is no vaccine against the virus and treatment is aimed only at easing symptoms. The virus survives the host’s immune response by means of a “cap” that protects its genomic RNA from enzyme degradation.
The cap is put in place by an enzyme, nonstructural protein 5 (NS5), which catalyzes a series of methyl transfer reactions on the RNA, but the precise details of this interaction have remained unclear. Pei-Yong Shi, Novartis Institute for Tropical Diseases, Singapore, Julien Lescar and Dahai Luo, Nanyang Technological University, Singapore, and colleagues present a crystal structure of full-length NS5, bound to the end of a viral RNA fragment.
This crystal structure is a snapshot of the methylation reaction at the 2′-O-ribose group of the first nucleotide in the sequence. It provides atomic-level information on the RNA-binding groove of NS5, which is useful for validating previous and future structural studies and also for designing inhibitors of this enzyme. It is hoped that interference with this methyl group transfer will form the basis of novel antiviral therapies against dengue fever.
- Molecular basis for specific viral RNA recognition and 2′-O-ribose methylation by the dengue virus nonstructural protein 5 (NS5),
Y. Zhao, T. S. Soh, S. P. Lim, K. Y. Chung, K. Swaminathan, S. G. Vasudevan, P.-Y. Shi, J. Lescar, D. Luo,
Proc. Natl. Acad. Sci. U.S.A. 2015.
DOI: 10.1073/pnas.1514978112