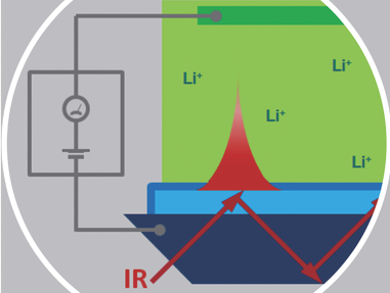
For many electrochemical systems, interfacial phenomena are of prime importance. Especially processes taking place on the solid/electrolyte interphase (SEI) are of interest. Unfortunately, techniques used so far only focus on the electrode material or its structure, but never on the changes on the SEI in its working status.
Francois Ozanam, CNRS-Ecole Polytechnique, Palaiseau, France, and colleagues used multiple internal reflection Fourier-transformed infrared spectroscopy to obtain information on the formation and evolution of the SEI on amorphous silicon lithium-ion electrodes during cycling. The team obtained information about the quantitative evolution of the SEI thickness and its chemical composition during cycling. Furthermore, they were able to identify the lithium incorporation in the electrode as the rate limiting step of the electrochemical process. The researchers detected distinct lithiation and delithiation processes during the first electrochemical cycle.
All these insights help to understand the behavior of the SEI better and to improve its properties, as additives to the electrolyte or a specific surface treatment may affect SEI properties and enhance long-term stability of the electrode.
- Spectroscopic Insight into Li-Ion Batteries during Operation: An Alternative Infrared Approach,
Daniel Alves Dalla Corte, Georges Caillon, Christian Jordy, Jean-Noël Chazalviel, Michel Rosso, François Ozanam,
Adv. Energy Mater. 2015.
DOI: 10.1002/aenm.201501768