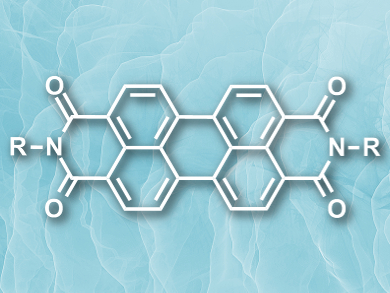
Developing rapid, compact, reaction-based alkane sensors is challenging because alkanes are relatively inert at room temperature. Ling Zang, University of Utah, Salt Lake City, USA, and colleagues designed an alkane sensor based on a composite film. The film is made of nanofibrils of donor and acceptor compounds with interlaced alkyl chains. The donor is a large alkyl-substituted arylene-ethynylene tetracycle, and the acceptor is perylene-3,4,9,10-tetracarboxylic-3,4,9,10-diimide (pictured), modified with dodecyl side chains.
Co-assembly of the two components produces a large surface contact area which optimizes interactions between the alkane and the sensor interface. Alkane adsorption increases the distance between the interlaced alkyl chains, reducing charge transfer between them and decreasing the associated photocurrent.
Short-chain alkanes reduce the photocurrent more than longer chains, and the photocurrent recovers more quickly for short-chain alkanes. Even at alkane vapor levels of 1 % of the saturation value, the signal was at least seven times larger than the background noise. Common polar solvents increase, rather than reduce, the photocurrent, providing a means to distinguish them from alkanes.
- Interfacial Donor–Acceptor Nanofibril Composites for Selective Alkane Vapor Detection,
Chen Wang, Benjamin R. Bunes, Miao Xu, Na Wu, Xiaomei Yang, Dustin E. Gross, Ling Zang,
ACS Sens. 2016.
DOI: 10.1021/acssensors.6b00018