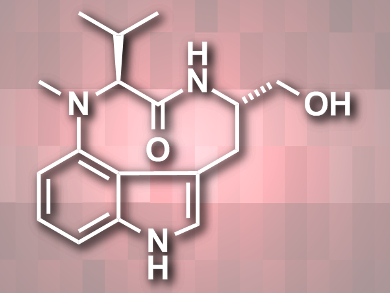
Protein kinase C (PKC) is an enzyme family important for cellular signal transduction. Drugs targeting these enzymes have been used for treatment of diseases such as cancer, Alzheimer’s, and HIV/AIDS. Indolactam alkaloids are a class of compounds that regulate the activity of PKC. They have been used to study, e.g., tumor growth and neurodegeneration. Systematic synthetic approaches to functionalized indolactams could further the understanding of their biological activity.
Kelvin L. Billingsley, San Francisco State University, CA, USA, and colleagues have synthesized the parent structure of these alkaloids, (−)-indolactam V (pictured), using a concise eight-step approach. The team started from a 4-bromoindole and used a copper-catalyzed amino acid arylation to couple 4-bromoindole with valine, converted the amino acid to a dipeptide, and finally closed the ring with an intramolecular indole alkylation.
According to the researchers, this modular synthesis allows the introduction of diverse hydrophobic groups by varying the amino acid reactants. The team is working on the synthesis and biological evaluation of a variety of indolactams.
- Modular Total Synthesis of Protein Kinase C Activator (−)-Indolactam V,
Jeremy Haynes-Smith, Italia Diaz, Kelvin L. Billingsley,
Org. Lett. 2016.
DOI: 10.1021/acs.orglett.6b00614