Sterically demanding and strongly electron donating ligands can stabilize compounds with unsaturated coordination centers or unusual redox states. N-heterocyclic carbenes (NHCs) are commonly used for this purpose, and have been reported to stabilize rare three-coordinate iron complexes. The related carbones, complexes of the type L2C, consist of two electron-donating ligands and one carbon atom with two free electron pairs. Carbodiphosphoranes (CDPs) with the general formula (PR3)2C, for example, are a strongly electron-donating type of carbone. However, no iron–CDP complexes had been reported thus far.
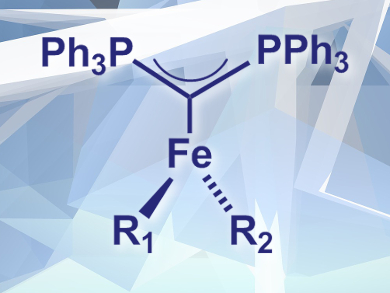
Douglas W. Stephan, University of Toronto, ON, Canada, and colleagues have synthesized the first CDP complexes of iron. The team prepared hexaphenylcarbodiphosphorane (PPh3)2C by deprotonation of the corresponding diphosphonium salt with K[N(SiMe3)2]. They combined the CDP with FeCl2(PPh3)2 in toluene, resulting in a three-coordinate iron complex (CDP)FeCl2 (pictured, R1 = R2 = Cl). The complex was found to exist in equilibrium with a four-coordinate dimer.
The resulting complexes were characterized using X-ray crystallography as well as 1H NMR and 57Fe Mössbauer spectroscopy. The researchers found that the complex undergoes ligand exchange with BnMgCl to give a dibenzyl derivative. Reactions with other nucleophiles were also observed. The prepared complexes are highly sensitive, however, the team hopes that the use of chelating ligands will result in similar, more stable species.
- Three and Four Coordinate Fe Carbodiphosphorane Complexes,
Conor Pranckevicius, Diana Alexandra Iovan, Douglas W Stephan,
Dalton Trans. 2016.
DOI: 10.1039/c6dt03453e