Electrochemically Controlled Calcium Release – Poster Presentation
Ferrocene Derivatives for Electrochemically Controlled Calcium Release
Ms Koyel X. Bhattacharyya, Ecole Normale Supérieure, Paris, France
We are interested in temporal and spatial control in cation delivery with minimal perturbation of biological systems. The controlled release of nanoquantities of ions plays a role in many cellular functions, such as insulin release and nerve impulses. The study of these functions requires the ability to monitor and regulate the ion flux externally. Traditional methods of ion flux control use micropipettes, which strongly perturb biological systems and synthesis of BAPTA derivatives.
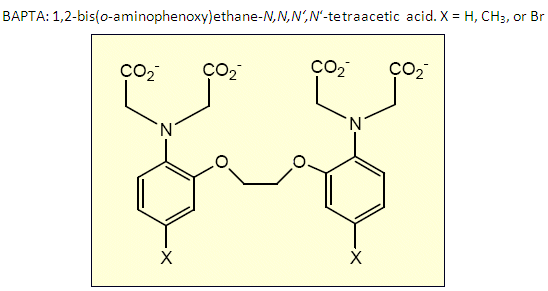
In order to examine biological properties such as oxidative stress and the vesicular release of neurotransmitters by inducing jumps in the concentration of cation effectors near a living cell, the existing aza-crown ether chemistry must be adapted for aqueous media, requiring the construction of ligands that form a stronger binding pocket for the cations, because water, a fairly strong ligand itself, competes with the ligands already in place, allowing the ions to diffuse into aqueous media at lower ion concentrations.
Here, we investigate the electrochemical control of ion concentrations from a distance, permitting more accurate regulation with minimal perturbation.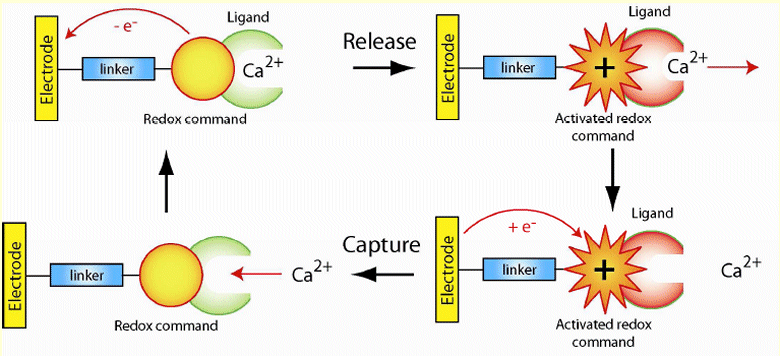
- Electrochemically Driven Release of Picomole Amounts of Calcium Ions with Temporal and Spatial Resolution
C. Amatore, D. Genovese, E. Maisonhaute, N. Raouafi, B. Schöllhorn,
Angew. Chem. Int. Ed. 2008, 47(28), 5211 – 5214.
DOI: 10.1002/anie.200705274 - C. Amatore, D. Genovese, E. Maisonhaute, N. Raouafi, B. Schöllhorn,
Angew. Chem. 2008, 120(28), 5289 – 5292.
DOI: 10.1002/ange.200705274



