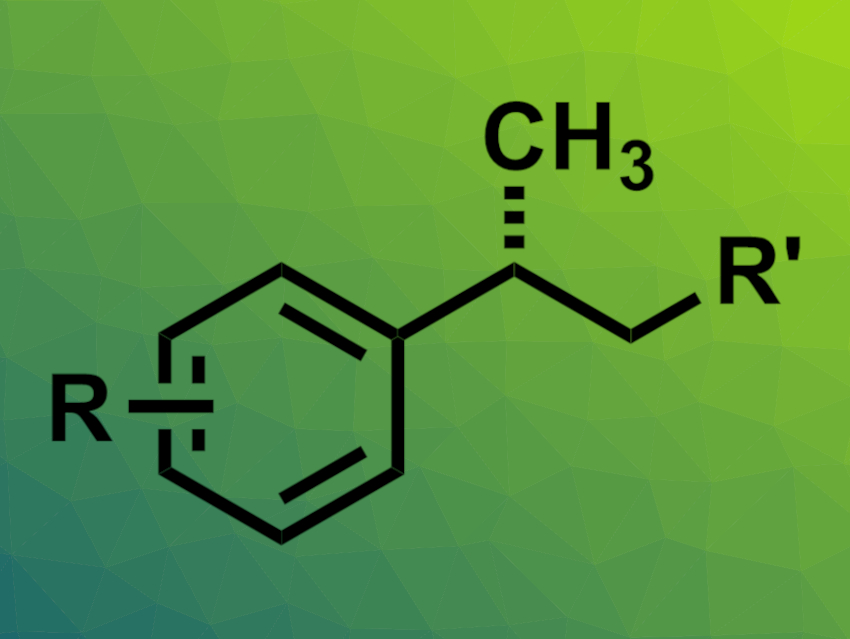
Adding a methyl group to an organic molecule can significantly change, e.g., its biological activity. Alkyl groups can be introduced, e.g., via asymmetric alkene functionalizations. However, approaches of this type for the direct installation of methyl groups are relatively rare. Asymmetric hydromethylations, for example, often need highly reactive methylating reagents and have a limited substrate scope.
Peng Liu, University of Pittsburgh, PA, USA, Stephen L. Buchwald, Massachusetts Institute of Technology (MIT), Cambridge, USA, and colleagues have developed a copper hydride-catalyzed enantioselective approach to the enantioselective hydromethylation of alkenes. The team avoided highly reactive, commonly used methyl sources such as methyl iodide and used methyl tosylate as a less reactive methyl source (MeOTs).
The researchers reacted a variety of styrene derivatives with MeOTs in the presence of sodium trimethylsilanolate (NaOTMS) and PhMe2SiH in tetrahydrofuran (THF) at 45 °C. As a catalyst, they used CuI together with (S)-DTBM-SEGPHOS as a bidentate chiral bisphosphine ligand.
In the reaction, the methyl tosylate is transformed in situ into methyl iodide by a catalytic amount of iodide. The team proposes a mechanism that involves a stereoselective hydrocupration, a stereoretentive methylation via an oxidative addition, and a reductive elimination to give the product. The desired hydromethylated products were obtained in moderate to good yields and with high enantioselectivities, and the method tolerates a broad range of functional groups.
- Copper Hydride-Catalyzed Enantioselective Olefin Hydromethylation,
Yuyang Dong, Kwangmin Shin, Binh Khanh Mai, Peng Liu, Stephen L. Buchwald,
J. Am. Chem. Soc. 2022.
https://doi.org/10.1021/jacs.2c07489