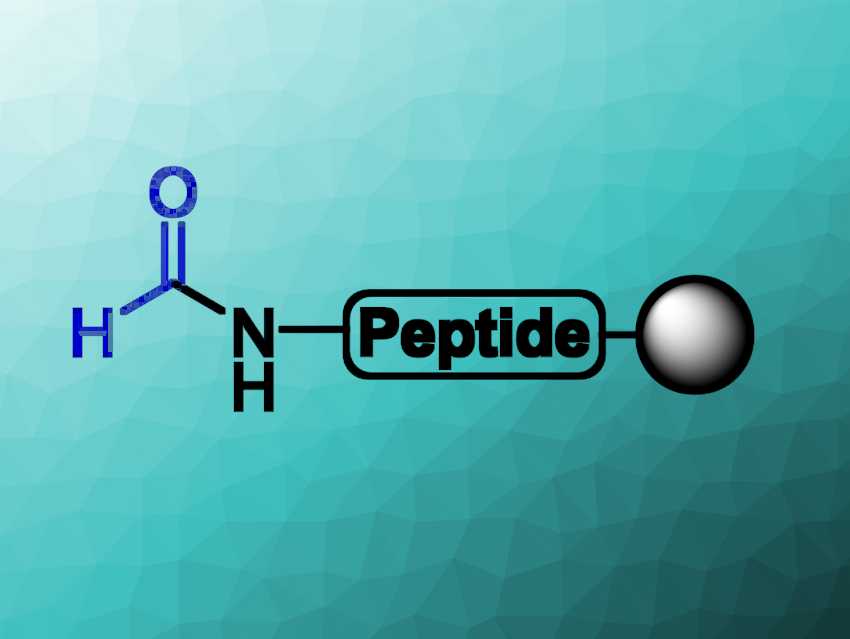
Modifications of peptides and proteins can strongly alter their biological effects. For example, the formylation of the N-terminus is typical for bacterial proteins and causes the proteins to be recognized by the human immune system. Another example is the N-formylation of lysine side chains of histone proteins in humans, which plays a role in epigenetics (the control of gene activity without changing the DNA sequence).
Truc Lam Pham, Franziska Thomas, University of Heidelberg, Germany, and colleagues have developed a simple and rapid method to N-formylate protected peptides on a resin directly after solid-phase synthesis, using readily available and shelf-stable reagents. The procedure involves swelling the peptide resin in dimethylformamide (DMF), followed by the addition of pyridine and formic acid. Acetic anhydride is then added under stirring over 10 min. Finally, the functionalized peptides were cleaved from the resin.
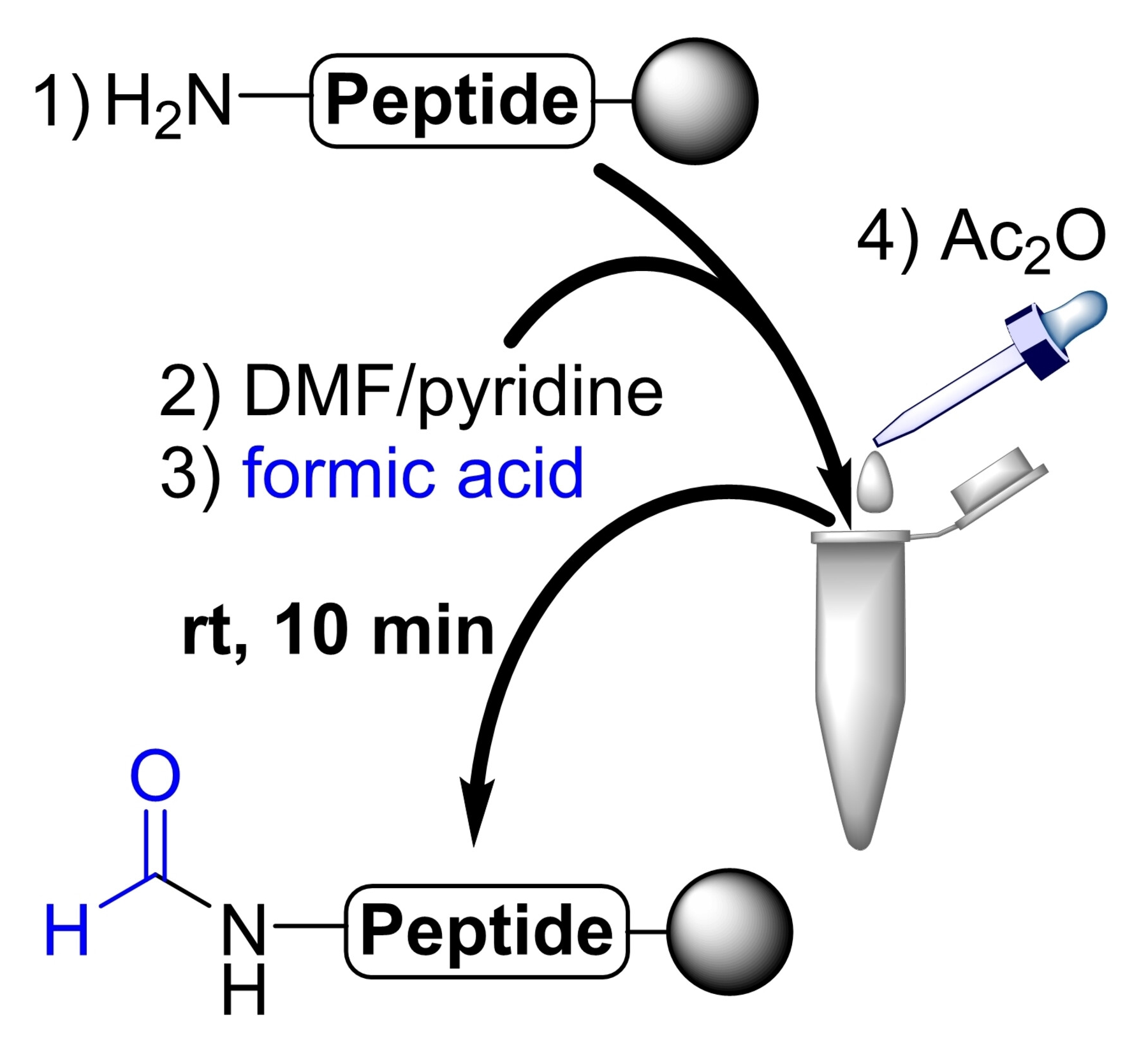
With this method, peptides can be formylated at the N-terminus as well as at lysine side chains with yields of over 90 %. This is independent of the peptide length and complexity—the method was shown to work for peptides with lengths of up to 34 amino acids and for the spectrum of canonical amino acids. Due to its simplicity, the approach could also be used by non-experts to produce formylated peptides.
- Rapid On‐Resin N‐Formylation of Peptides as One‐Pot Reaction,
Agon Kokollari, Marius Werner, Christina Lindner, Truc Lam Pham, Franziska Thomas,
ChemBioChem 2023.
https://doi.org/10.1002/cbic.202300571