Biological macromolecules such as proteins are usually characterized in vitro after their removal from living cells. Studying them in cells instead could be useful to fully understand their biological functions. In-cell NMR spectroscopy is useful for this. However, it is subject to certain limitations such as signal loss due to line-broadening effects in the cellular environment. This line broadening is due to interactions between the target molecule and other abundant compounds and adversely affects 1H NMR spectra.
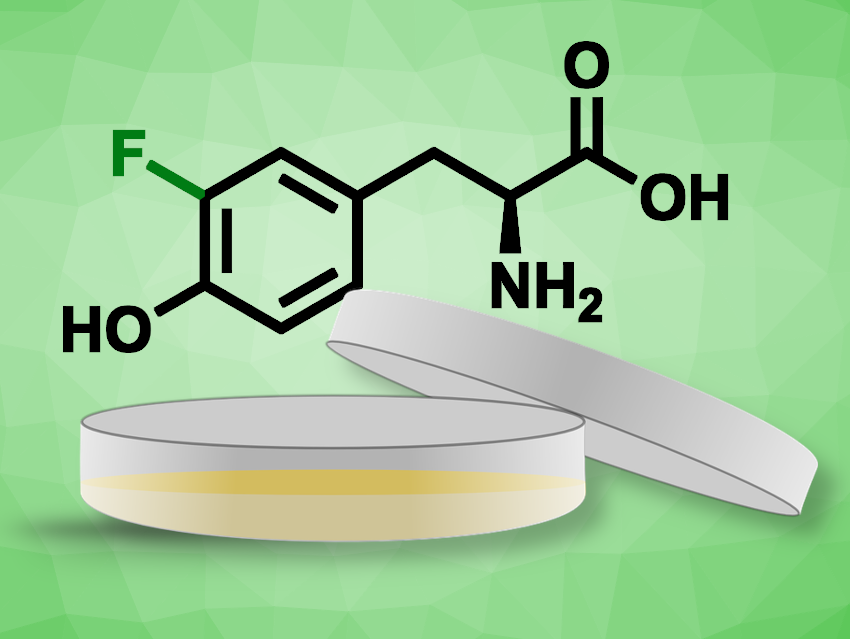
19F NMR spectroscopy can overcome this limitation due to the absence of 19F in the cell environment, which leads to spectra with significantly less background interference. Usually, proteins labeled with fluorinated amino acids are produced in bacteria and either analyzed directly in bacterial cells or delivered into human cells. Methods for direct protein expression in human cells using fluorinated amino acids would be useful.
Enrico Luchinat, Consorzio Interuniversitario Risonanze Magnetiche di Metallo Proteine (CIRMMP), Sesto Fiorentino, Italy, Alma Mater Studiorum – Università di Bologna, Cesena, Italy, Lucia Banci, Università degli Studi di Firenze and CIRMMP, Sesto Fiorentino, Italy, and colleagues have found that several fluorinated aromatic amino acids can be easily incorporated in proteins expressed in human cells. The team achieved this by replacing an amino acid in the cells’ growth medium with the corresponding fluorinated amino acid during the incubation of the cells.
The researchers achieved fluorination efficiencies of up to 60 %. The target proteins were successfully detected in 19F in-cell NMR spectra. According to the team, this is the first in-cell 19F NMR application where proteins are expressed and fluorinated directly in human cells.
- Direct Expression of Fluorinated Proteins in Human Cells for 19F In-Cell NMR Spectroscopy,
Lan B. T. Pham, Azzurra Costantino, Letizia Barbieri, Vito Calderone, Enrico Luchinat, Lucia Banci,
J. Am. Chem. Soc. 2023.
https://doi.org/10.1021/jacs.2c12086