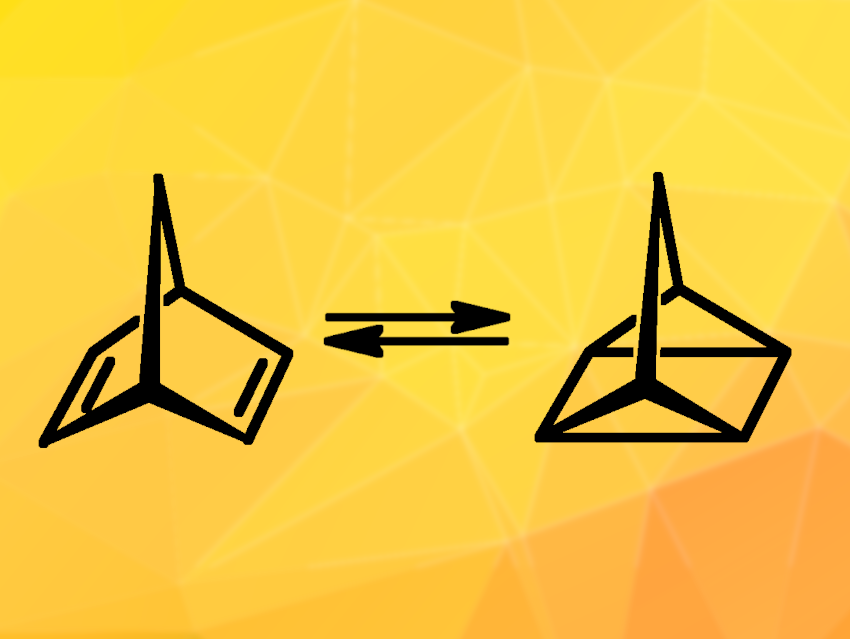
The storage of solar energy is important to improve the use of renewable energy. One approach to the storage of solar energy is the use of molecular solar thermal (MOST) systems. They are based on molecular switches, which isomerize under sunlight into a high-energy state. The stored energy can later be released on demand as heat. The photoswitch, thus, combines conversion, storage, and release functionalities in a single molecule.
Olaf Brummel, University of Erlangen-Nuremberg, Erlangen, Germany, and colleagues have investigated a prominent MOST system based on a norbornadiene/quadricyclane photoswitch (NBD/QC, pictured). To initiate the energy storage process, the NBD derivative cyano-3-(3,4-dimethoxyphenyl)-norbornadiene (NBD′) is converted to its energy-rich, metastable photoisomer, a QC derivative (QC’), upon exposure to sunlight. The energy release process then involves the conversion of QC’ to NBD’. Controlling this back-conversion is difficult—a major challenge for the application of such MOST systems.
The team investigated what happens when the energy release is heterogeneously catalyzed over an Au(111) surface. The system was studied using photoelectrochemical infrared reflection absorption spectroscopy (PEC-IRRAS) and scanning tunneling microscopy (STM).
The researchers found that the Au catalyst provides high activity, leading to high heat release rates. The release can be controlled by application of external potential. This could allow control over the energy release rate in future MOST devices in a straightforward manner. The system also shows high stability over 1,000 storage cycles, with an activity decrease of just 0.1 % per cycle.
- Tunable Energy Release in a Reversible Molecular Solar Thermal System,
Evanie Franz, Corinna Stumm, Fabian Waidhas, Manon Bertram, Martyn Jevric, Jessica Orrego-Hernández, Helen Hölzel, Kasper Moth-Poulsen, Olaf Brummel, Jörg Libuda,
ACS Catal. 2022.
https://doi.org/10.1021/acscatal.2c03043