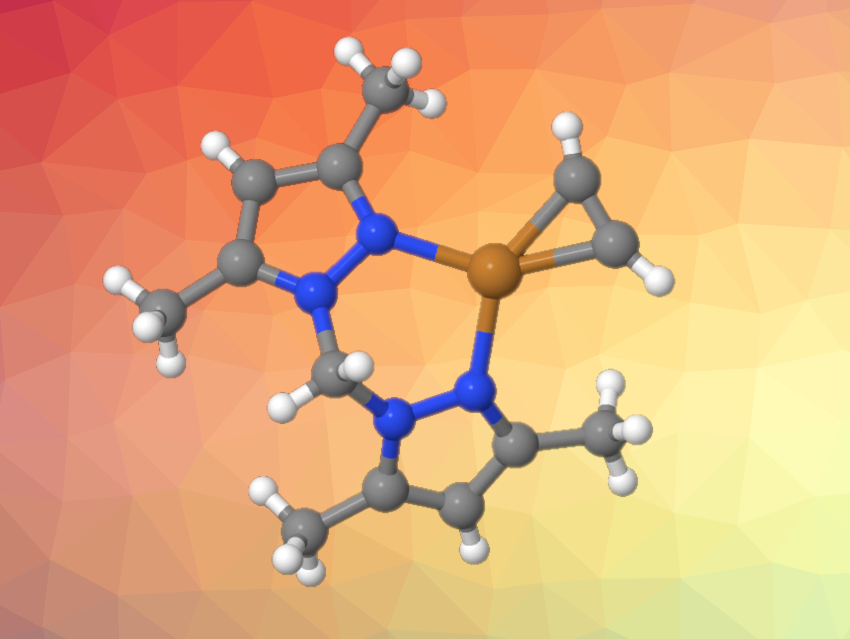
Acetylene (C2H2) is an important building block in industrial chemistry and organic synthesis. Silver and copper complexes can be useful in catalytic transformations of acetylene, but isolable molecules in which acetylene is bound to Cu(I) or Ag(I) centers are rare.
Israel Fernández, Universidad Complutense de Madrid, Spain, Rasika Dias, The University of Texas at Arlington, USA, and colleagues have isolated several new acetylene adducts of Cu(I) and Ag(I), i.e., acetylene complexes supported by pyrazole-based chelating ligands (example pictured). The team used the fluorinated tris(pyrazolyl)borates [HB(3,5-(CF3)2Pz)3]− and [HB(3-(CF3)-5-(Ph)Pz)3]−, the fluorinated bis(pyrazolyl)borate ligand [Ph2B(3-(CF3)Pz)2]−, and the bis(pyrazolyl)methane ligand H2C(3,5-(CH3)2Pz)2. The complexes were formed readily in solution under an atmosphere of excess acetylene from corresponding precursor complexes.
The team fully characterized the products using X-ray crystallography as well as 1H, 13C, and 19F NMR and Raman spectroscopy. They also performed density functional theory (DFT) calculations to investigate the bonding situation in the complexes. They found that there is a stronger interaction between the transition-metal fragment and the acetylene in the Cu(I) complexes than in the Ag(I) compounds. According to the researchers, the products represent the largest collection
of isolable Cu(I) and Ag(I) complexes featuring a terminal, η2-HC≡CH ligand.
- Isolable acetylene complexes of copper and silver,
Rasika Dias, Anurag Noonikara Poyil, Shawn G. Ridlen, Israel Fernandez,
Chem. Sci. 2022.
https://doi.org/10.1039/d2sc02377f