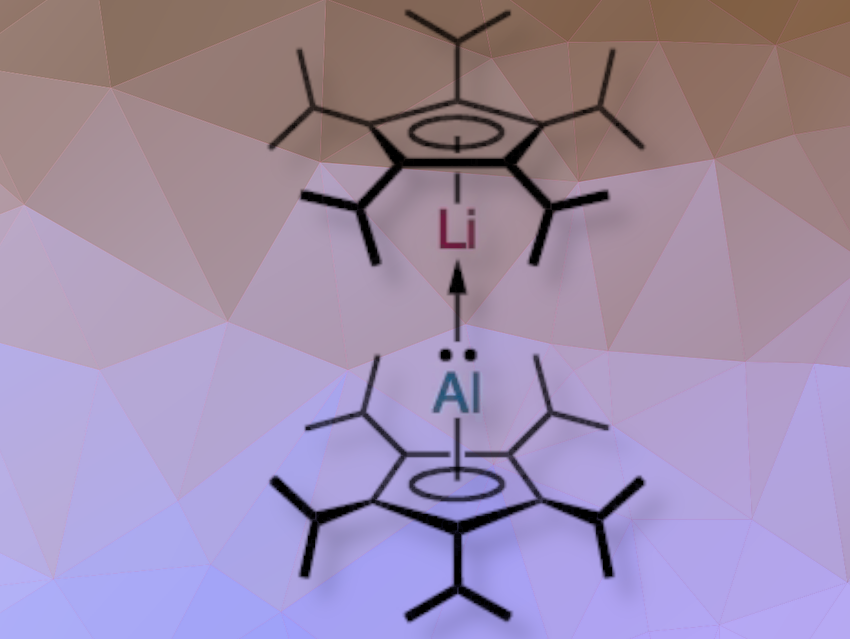
André Schäfer and colleagues, Saarland University, Saarbrücken, Germany, have synthesized a heterobimetallic dimetallocene by heterocoupling of lithium and aluminylene fragments with pentaisopropylcyclopentadienyl ligands. The term dimetallocene refers to very rare sandwich complexes in which two metal atoms are bonded between the η5-coordinated cyclopentadienyl (Cp) ligands arranged in linear/coplanar fashion and interlinked by a metal–metal bond. Dimetallocenes of p-block elements were so far unknown.
The team first synthesized (5Cp)aluminylene (cyclopentadienylaluminylenes) using (5Cp)lithium diethyletherate, 5CpLi∙OEt2, and the (pentamethylcyclopentadienyl)aluminium(I) tetramer as a precursor. The ability of (5Cp)aluminylene to act as a donor ligand despite the bulky 5Cp group makes it an excellent candidate for the targeted synthesis of a heterobimetallic dimetallocene.
The team then reacted it with an equivalent of 5CpLi in the presence of Cp*Li as diethyl ether scavenger, resulting in the formation of the lithium–aluminium heterobimetallic dimetallocene. The Al–Li bond has a strong ionic nature and benefits from attractive dispersion interactions between the isopropyl groups of the cyclopentadienyl ligands. Because the Al–Li bond is relatively weak, it is cleaved easily by donor molecules such as an N-heterocyclic carbene (NHC) or in reactions with heteroallenes, such as phenylisocyanate, mesitylisothiocyanate, and 1-azidoadamantane. These reactions led to the formation of (5Cp)lithium complexes and dialumazine.
Lithium–aluminium dimetallocene is formally valence-isoelectronic to dizincocene and diberyllocene, but the cleavage of the Al–Li bond is in sharp contrast to the related valence-isoelectronic dizincocene, in which the Zn–Zn bond is perpetuated upon coordination of an NHC.
- A lithium–aluminium heterobimetallic dimetallocene,
Inga-Alexandra Bischoff, Sergi Danés, Philipp Thoni, Bernd Morgenstern, Diego M. Andrada, Carsten Müller, Jessica Lambert, Elias C. J. Gießelmann, Michael Zimmer, André Schäfer,
Nature Chem. 2024.
https://doi.org/10.1038/s41557-024-01531-y




Thank you very much for highlighting our work!
AS
https://www.uni-saarland.de/fakultaet-nt/schaefer.html