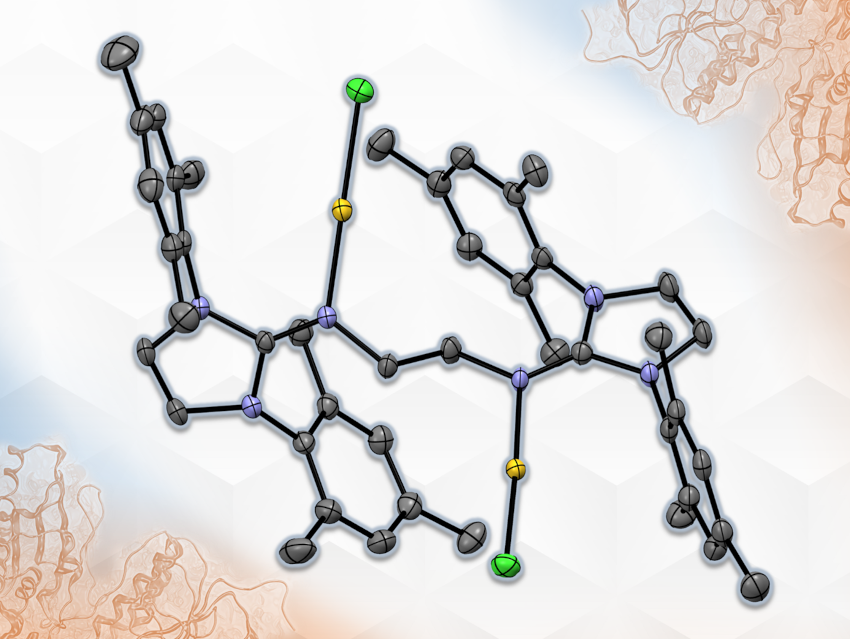
The discovery of the anticancer effect of gold compounds, whose mode of action differs from that of Pt(II) chemotherapeutics, has fuelled research toward their design and optimization as targeted therapeutics. A major challenge for clinical use is controlling the speciation of metallodrugs in biological environments. This means that the development of new ligands for enhanced complex stability would be useful. N-heterocyclic imines (NHIs) are interesting in this context as highly electron-donating ligands. While NHI complexes have been investigated for their catalytic activities, their medicinal use has so far remained unexplored.
Angela Casini, Shigeyoshi Inoue, Technical University of Munich, Garching, Germany, and colleagues have synthesized the dinuclear bis-NHI Au(I) complex 1,2-(IMesN-AuCl)2-C2H4 (pictured, IMes = 1,3-bis(mesityl)imidazolin-2-ylidene) and tested its antiproliferative effects against a panel of human cancer cell lines in vitro. The complex was synthesized from AuCl(SMe2) and the NHI ligand via a ligand exchange reaction at room temperature.
The complex showed good stability under aqueous conditions. The team found that the complex is more potent and selective against A549 non-small cell lung cancer (NSCLC) cells than the benchmark metallodrugs auranofin and cisplatin. Initial studies of the compound’s mode of action identified the seleno-enzyme thioredoxin reductase, responsible for maintaining the intracellular redox balance, as a likely target in A549. Overall, the work shows the potential of complexes with NHI ligands for medicinal applications.
- Potent Anticancer Activity of a Dinuclear Gold(I) Bis‐N‐Heterocyclic Imine Complex Related to Thioredoxin Reductase Inhibition in Vitro,
Mihyun Park, Claudia Schmidt, Sebastian Türck, Franziska Hanusch, Simone V. Hirmer, Ingo Ott, Angela Casini, Shigeyoshi Inoue,
ChemPlusChem 2023.
https://doi.org/10.1002/cplu.202300557