Several vaccines against COVID-19 have proven safe and effective in clinical trials. mRNA-based vaccines are widely used, but there is also a protein-based COVID-19 vaccine. Protein-based vaccines can be stored and produced fairly easily, but often need adjuvants to enhance their effect. Adjuvants are added compounds that help vaccines to elicit a more robust immune response.
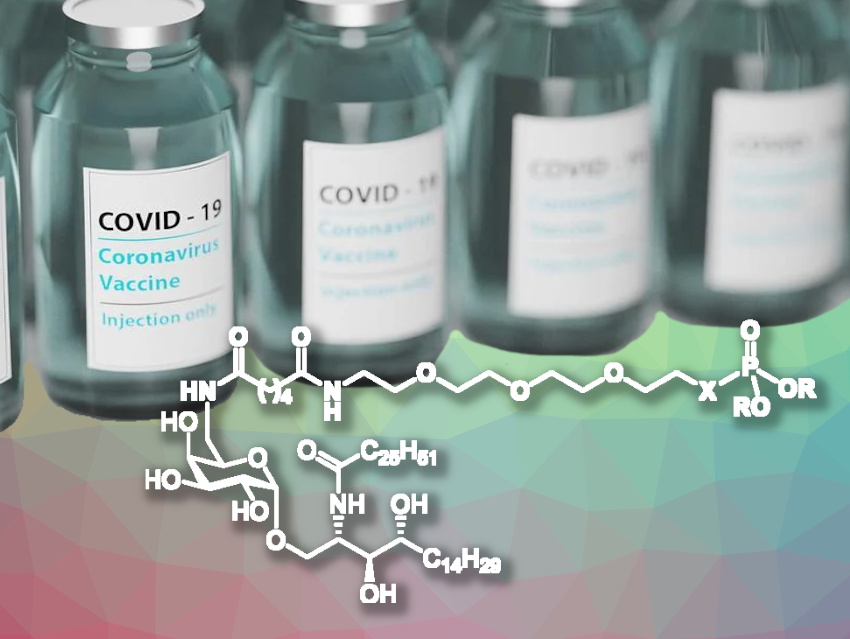
Rui Luo, Huazhong Agricultural University, Wuhan, China, Zheng Liu, Central China Normal University, Wuhan, China, and colleagues have found that analogues of α-galactosylceramide (αGC, analogues pictured) can act as adjuvants. αGC stimulates invariant natural killer T (iNKT) cells, a type of immune cell that is important for mediating strong and rapid responses to a range of diseases. The team synthesized four analogs of αGC and added them separately to a vaccine based on the receptor-binding domain (RBD) of the SARS-CoV-2 spike protein. In the analogues, αGC is covalently bound via polyethylene glycol (PEG) linkers to either a phosphate, a phosphonate, a difluoromethylenephosphonate, or a phosphonate diester group.
The resulting adjuvanted vaccines were tested in mice. The animals received three injections over the course of 29 days and their immune response was tracked. The phosphonate diester analogue called αGC-CPOEt (X = CH2, R = CH2CH3) led to the production of antibodies with the greatest neutralizing capacity—5.5 times greater than with αGC itself and 25 times greater than without an adjuvant. These results suggest that αGC-CPOEt merits further investigation as a potential adjuvant.
- A New iNKT-Cell Agonist-Adjuvanted SARS-CoV-2 Subunit Vaccine Elicits Robust Neutralizing Antibody Responses,
Ya-Qian Li, Cheng Yan, Xi-Feng Wang, Mao-Ying Xian, Guo-Qing Zou, Xiao-Fei Gao, Rui Luo, Zheng Liu,
ACS Infect. Dis. 2022.
https://doi.org/10.1021/acsinfecdis.2c00296
Also of Interest
- Collection: SARS-CoV-2 Virus
What we know about the new coronavirus and COVID-19 - Clever Picture: Comparing COVID-19 Vaccines,
ChemistryViews 2021.
https://doi.org/10.1002/chemv.202100033
We look here at a few of the differences between the EU-approved vaccines.