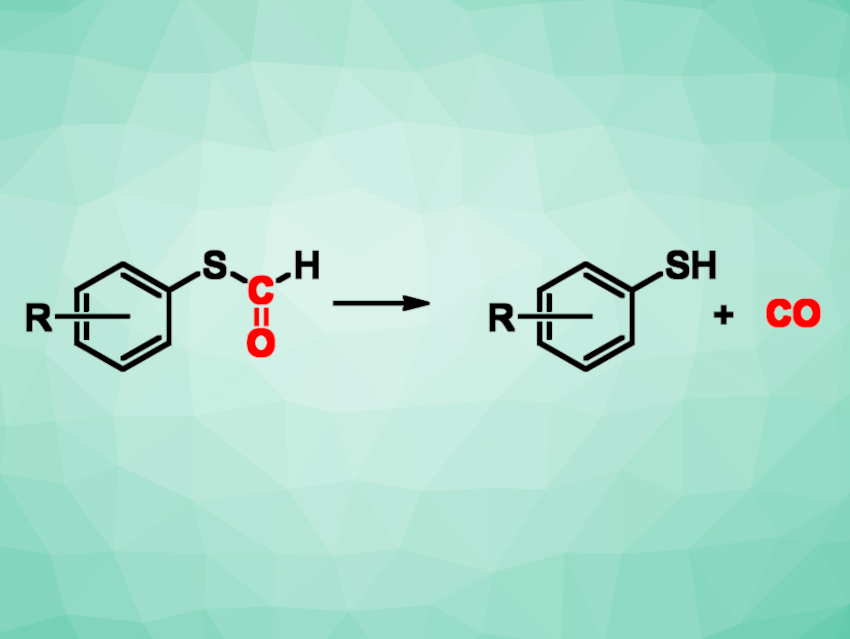
Carbon monoxide (CO) is widely used in chemistry, e.g., in organic synthesis or inorganic complexes. However, CO is toxic and a gas cylinder of CO in a laboratory is a potential safety hazard, as well as impractical when only very small amounts of CO are needed. CO-releasing molecules (CORMs) can provide a useful alternative approach for generating small amounts of CO on demand.
Sarah Z. Tasker, Franklin & Marshall College, Lancaster, PA, USA, and colleagues have discovered tunable and cost-effective CORMs. While studying the photochemistry of S-aryl thioformates, the team noticed an unexpected bubbling upon addition of an amine. The formed gas was CO, which was quantitatively produced at room temperature by adding an amine like iPr2EtN to the thioformate—which the team then termed a “thioCORMate”.
The “thioCORMates” offer advantages over existing CORMs. They are bench-stable, can be easily synthesized from the corresponding thiophenol in one step, and can be electronically tuned to release CO at different rates by changing the substituents on the aryl ring. In addition, a 13C-thioCORMate synthesized using isotopically labeled formic acid can be used as a convenient source of 13CO gas. According to the researchers, thioCORMates could provide CO for a variety of uses, including carbonylative cross-coupling reactions, inorganic synthesis, and biological studies.
- ThioCORMates: Tunable and Cost‐Effective Carbon Monoxide‐Releasing Molecules,
Catherine A. DeSimone, S. Lyla Naqvi, Sarah Zinnen Tasker,
Chem. Eur. J. 2022.
https://doi.org/10.1002/chem.202201326