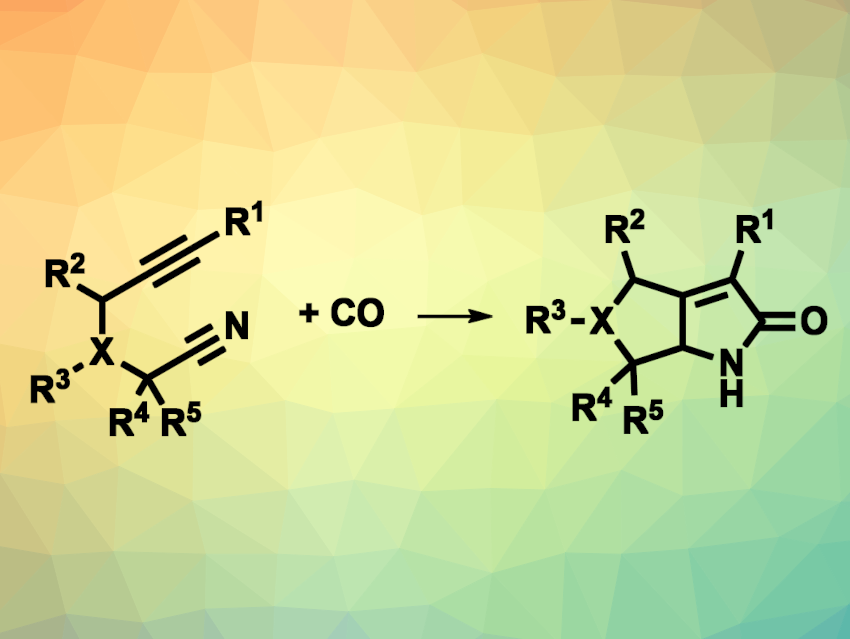
The Pauson–Khand reaction (PKR) is a [2+2+1] cycloaddition between an alkyne, an alkene, and carbon monoxide. Variations of this type of reaction that involve heteroatoms could be useful for the synthesis of five-membered heterocycles. γ-Lactams, for example, are cyclic amides with a five-membered ring. They are often found in pharmaceutically active compounds or natural products. Aza-Pauson-Khand reactions can be useful for the synthesis of γ-lactams, but examples that use nitrile groups are rare.
Wen-Bo Liu, Wuhan University, China, and Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences, and colleagues have developed a reductive aza-Pauson-Khand reaction between an alkyne group, a nitrile group, and carbon monoxide in the form of Co2(CO)8 that gives bicyclic α,β-unsaturated γ-lactams (general reaction pictured; X = C,O,N). The team combined a range of alkyne-functionalized nitriles with Co2(CO)8 in toluene at 80 °C, followed by removal of the toluene and the addition of N-methyl morpholine N-oxide (NMO) as a promoter in a MeCN/H2O solvent mixture at 60 °C.
The team obtained the desired bicyclic α,β-unsaturated γ-lactams in moderate to good yields and with high diastereoselectivities. When D2O was used as the cosolvent instead of H2O, deuterium incorporation at the γ-position of the products was observed. According to the researchers, the developed reaction offers an efficient pathway for [2+2+1] cycloadditions that are otherwise difficult to accomplish.
- Reductive Aza-Pauson-Khand Reaction of Nitriles,
Hong-Gui Huang, Yu-Qing Zheng, Dayou Zhong, Jiang-Lian Deng, Wen-Bo Liu,
J. Am. Chem. Soc. 2023.
https://doi.org/10.1021/jacs.3c01656