Ethylene is an important chemical feedstock, especially for the production of polyethylene. However, commercially produced ethylene generally has acetylene impurities, which can poison the commonly used polymerization catalysts, and thus, need to be removed. The selective hydrogenation of acetylene to ethylene can be used to solve this problem, but existing methods for this generally require high temperatures. A catalyst that promotes acetylene semi-hydrogenation at room temperature and atmospheric pressure with a high selectivity would, thus, be useful.
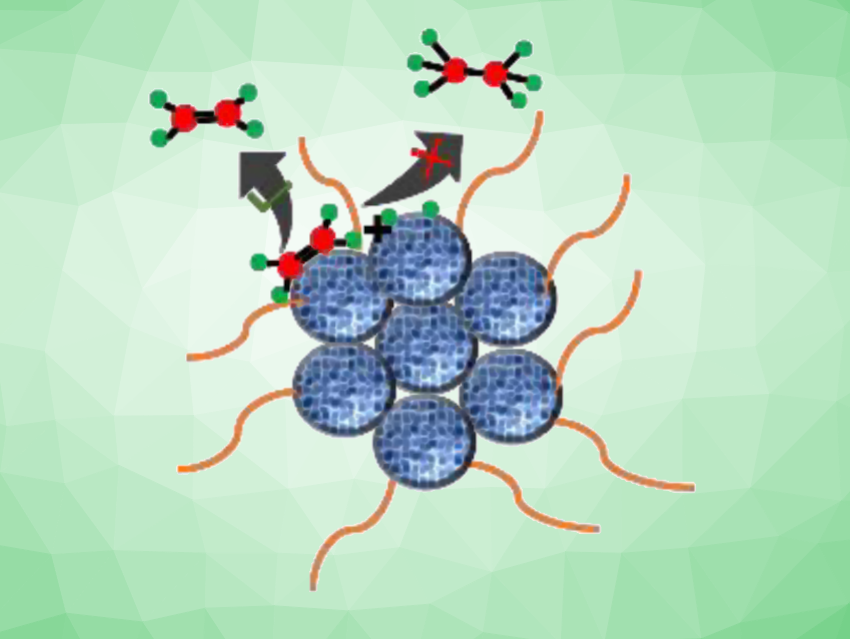
Vivek Polshettiwar, Tata Institute of Fundamental Research, Mumbai, India, Balaji R. Jagirdar, Indian Institute of Science, Bangalore, and colleagues have developed an approach to the semi-hydrogenation of acetylene to ethylene over hexadecylamine (HDA)-capped bimetallic Pd-Zn nanoparticles at room temperature that achieves high selectivity. The team synthesized palladium and zinc nanoparticles using a solvated metal atom dispersion (SMAD) approach and then carried out co-digestive ripening of these nanoparticles in mesitylene in the presence of hexadecylamine to form the desired bimetallic nanoparticles.
The researchers investigated whether the bimetallic nanoparticles can be used for the semi-hydrogenation of acetylene at room temperature and atmospheric pressure. The catalyst showed a high selectivity of ca. 85 % towards ethylene with a high ethylene productivity of about 4341 μmol g–1min–1. It also showed high stability, with an ethylene selectivity that stayed above 85 % even after 70 h on stream. The work could have applications in the polymer industry.
- Acetylene Semi‐hydrogenation at Room Temperature over Pd‐Zn Nanocatalyst,
Garima Tiwari, Gunjan Sharma, Rishi Verma, Pooja Gakhad, Abhishek Kumar Singh, Vivek Polshettiwar, Balaji Rao Jagirdar,
Chem. Eur. J. 2023.
https://doi.org/10.1002/chem.202301932