Self-assembled cages that can be used for host-guest chemistry are interesting, e.g., for the separation of molecules, catalysis, or molecular recognition. Building multiple functionalities into a single cage using different building blocks could be useful. However, using a variety of building blocks can make it challenging to avoid a random, statistical assembly of the components leading to a mixture of different products.
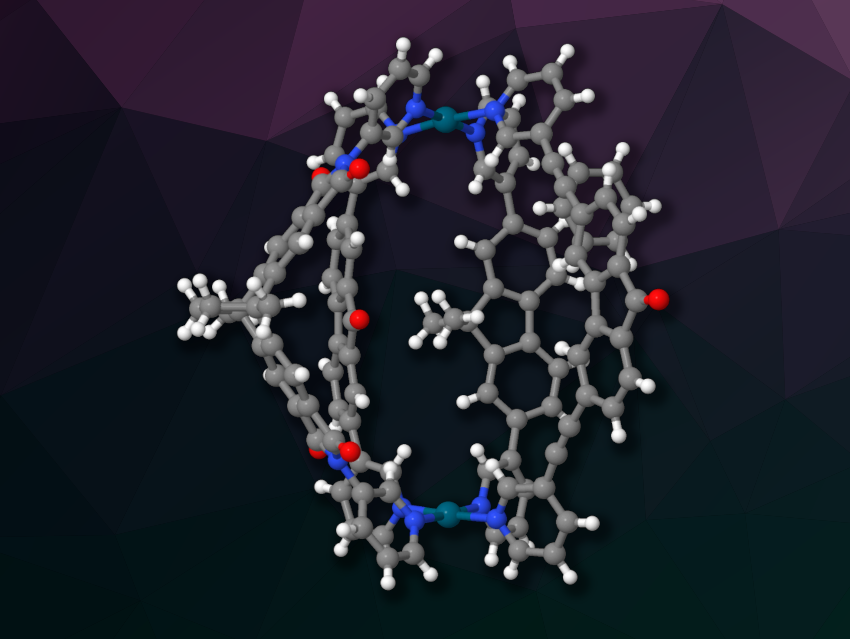
Guido H. Clever, TU Dortmund University, Germany, and colleagues have synthesized self-assembled cages in which two Pd(II) centers with square-planar coordination environments are bridged by four different bis-pyridyl ligands, giving a single isomer of a lantern-shaped [Pd2ABCD]4+ cage (pictured; Pd in dark green; A, B, C, and D = ligands). The team points out that, theoretically, 55 different species could be formed from the four different ligands and the two Pd centers. Nevertheless, they observed the exclusive formation of a heteroleptic [Pd2ABCD]4+ cage when they mixed the ligands A, B, C, and D with a Pd(II) species in a 1:1:1:1:2 ratio in CD3CN at 80 °C.
The assembly proceeds under thermodynamic control, and the products are formed in a reproducible manner. According to the researchers, this self-sorting effect is due to a combination of complementary ligand shapes and a balance of strain and C–H⋯π interactions between the ligands. For example, the geminal methyl groups on ligand C point to the π-surface of ligand D, forming C–H⋯π interactions. For the ribbon-shaped, flexible ligand A, the team proposes that exactly one “copy” of the ligand per cage is optimal to achieve the required length in combination with the other three ligands. Overall, the work could provide useful insights for the development of multifunctional self-assembled species.
- Non-statistical assembly of multicomponent [Pd2ABCD] cages,
Kai Wu, Elie Benchimol, Ananya Baksi, Guido H. Clever,
Nat. Chem. 2024.
https://doi.org/10.1038/s41557-023-01415-7