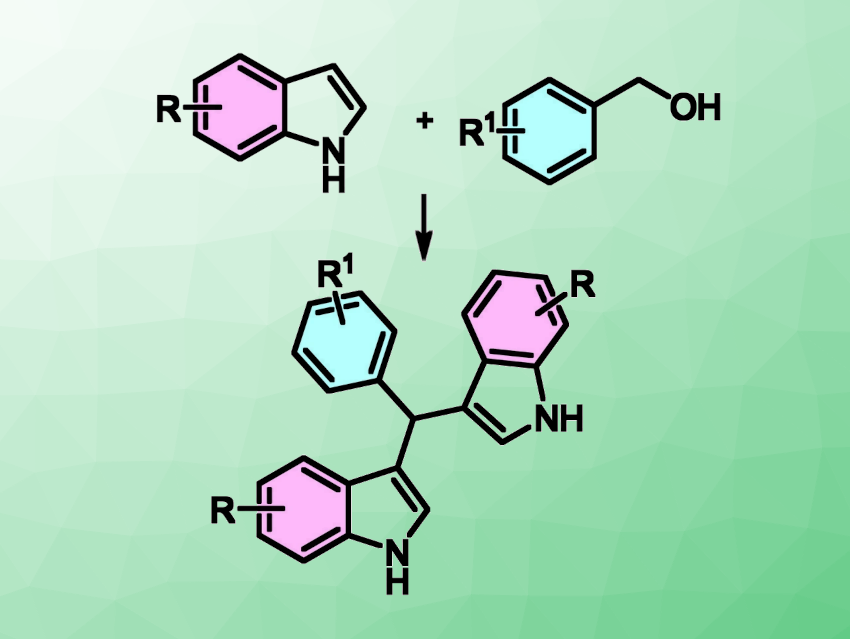
Bis(3-indolyl)methanes (BIMs) are compounds that contain two indole units (example pictured above in the bottom row). BIMs are important indole derivatives. They are found, e.g., in natural products and have potential applications in medicinal chemistry. BIMs have been synthesized by various methods, often using metal catalysis.
Manuel B. Marques, NOVA School of Science and Technology, Universidade Nova de Lisboa, Caparica, Portugal, and colleagues have developed a simple synthetic method that provides access to bis(3-indolyl)methanes (BIMs). The team used a tBuOK-mediated route (pictured) to prepare BIM derivatives from benzyl alcohols and indoles in yields of up to 77 %. The reactions were performed in toluene at 110 °C.
This simple method provides access to varied products (ten examples, including azaindole derivatives) in good yields. The team proposes that a radical mechanism is involved in the C-3 alkylation of indoles and azaindoles. According to researchers, the work can serve as a proof of concept for the relevance of tBuOK in C–C bond-forming reactions involving benzyl moieties.
- Synthesis of Bis(3‐indolyl)methanes Mediated by Potassium tert‐Butoxide,
A. Sofia Santos, Rita D. Ferro, Nuno Viduedo, Luísa B. Maia, Artur M. S. Silva, M. Manuel B. Marques,
ChemistryOpen 2023.
https://doi.org/10.1002/open.202200265


![A Path to Substituted Bicyclo[2.1.1]hexanones](https://www.chemistryviews.org/wp-content/uploads/2024/10/1substitutedbicyclo211hexan2ones_2024-125x94.png)
