The attachment of a phosphate unit to biomolecules is universally found in biochemistry. Much less well known is the biochemistry of phosphonates, in which the phosphorus forms a covalent bond with a carbon atom and which is mainly known from marine microbes, but rarely seen in land-based species. Isabelle Marcotte, Université du Québec à Montréal, Canada, Matthew J. Harrington, McGill University, Montreal, Canada, and colleagues were, therefore, surprised to discover that velvet worms use phosphonates to form a biological glue—sticky fibers formed from a special kind of slime [1].
The Velvet Worm’s Biological Glue
The velvet worm is an invertebrate a few centimeters in length that lives in humid habitats in the southern hemisphere. It catches its prey by shooting threads of sticky slime at it, which instantly harden into strong and sticky fibers that the prey cannot get rid of. Unlike other natural glues, however, the hardening is not permanent, and the fibers diss olve when water is added. This clever mechanism allows the velvet worm to fully recycle its sticky proteins into new slime and could have important implications for bioinspired materials research, especially for the identification of new recyclable and sustainable adhesive systems.
olve when water is added. This clever mechanism allows the velvet worm to fully recycle its sticky proteins into new slime and could have important implications for bioinspired materials research, especially for the identification of new recyclable and sustainable adhesive systems.
The sticky fibers are made from high-molecular-weight proteins, which are densely packed inside nanoglobules within the slime and assemble into fibrils when the nanoglobules dry out and collapse. When shear stress is applied, the fibrils harden into strings that are as tough as nylon fibers. Because electrostatic interactions, rather than covalent bonds, drive the protein assembly, incoming water can loosen the bonds between the proteins and dissolve the fibers back into their building blocks.
Phosphonates, Not Phosphates
Since large amounts of phosphorus were found in the slime, too, the team used NMR spectroscopy to investigate the role of phospholipids, which were thought to form the boundaries of the nanoglobules. The result of 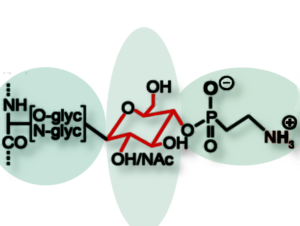 the analysis came as a surprise: “Instead of phosphorylation, we found phosphonates, a rare kind of molecule in terrestrial organisms,” says Alexandre Poulhazan, who is first author of the research article. “Phosphonates were unexpected, but the NMR data was unambiguous.”
the analysis came as a surprise: “Instead of phosphorylation, we found phosphonates, a rare kind of molecule in terrestrial organisms,” says Alexandre Poulhazan, who is first author of the research article. “Phosphonates were unexpected, but the NMR data was unambiguous.”
This analysis, supplemented with mass spectrometry, revealed that the phosphorus was present in a (2-aminoethyl)phosphonate moiety bound to glycans, which were attached to the large proteins that form the sticky fibers when exposed to air and shear stress. In questioning the role that the phosphonates might play in fiber formation, the researchers propose that their ability to form zwitterions helps to rapidly cross-link the proteins through electrostatic interactions.
A Rare Kind of Molecule in Terrestrial Organisms
To date, phosphonates have rarely been found in other land-bound species. However, they are common in marine microorganisms, where they appear to provide an evolutionary advantage over “normal” phosphorylation. For the velvet worm, a very ancient species of soft-bodied terrestrial invertebrates that have existed on Earth for several hundred million years, the team not only discovered phosphonates in the slimes of very distantly related species, but they also detected phosphonates in various parts of the worm’s body.
Given this peculiar presence of phosphonates in a non-marine organism, the researchers recommend that their role be investigated further and that phosphonates be considered “as a potential source of organophosphorus in understudied groups of animals and their biological functions.”
Reference
[1] Alexandre Poulhazan, Alexander Baer, Gagan Daliaho, Frederic Mentink-Vigier, Alexandre A. Arnold, Darren C. Browne, Lars Hering, Stephanie Archer-Hartmann, Lauren E. Pepi, Parastoo Azadi, Stephan Schmidt, Georg Mayer, Isabelle Marcotte, Matthew J. Harrington, Peculiar Phosphonate Modifications of Velvet Worm Slime Revealed by Advanced Nuclear Magnetic Resonance and Mass Spectrometry, J. Am. Chem. Soc. 2023, 145, 20749–20754. https://doi.org/10.1021/jacs.3c06798



