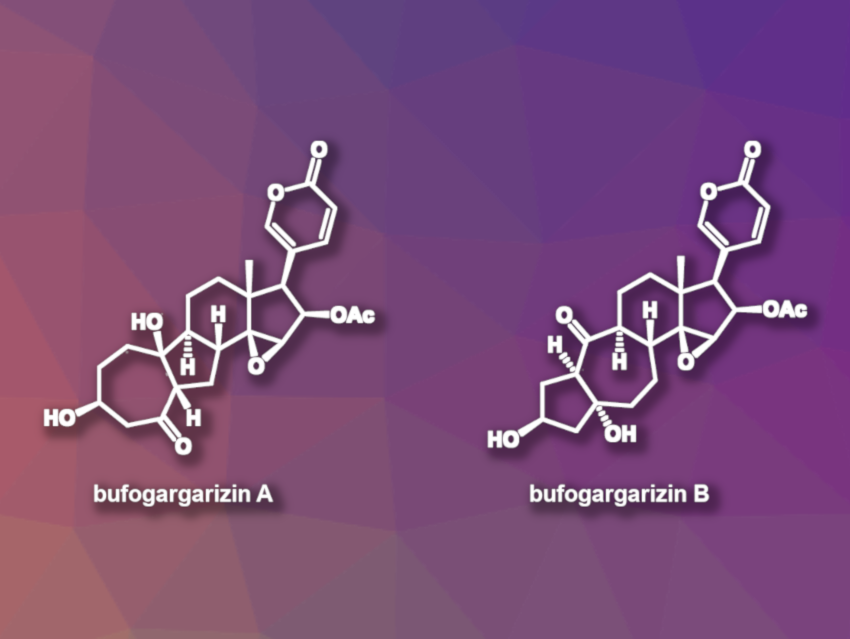
Steroids are compounds that often show biological activities and can be useful, e.g., in drug discovery. Bufogargarizins A and B (pictured) (Figure 1) are steroids with uncommon structures. The compounds contain unusual [7–5–6–5] and [5–7–6–5] tetracyclic skeletons, respectively. They were isolated from toad venom, which has been used in traditional Chinese medicine. In-depth studies of the biological activities of bufogargarizins A and B, however, have been hampered by the small amounts of the compounds available in Nature.
Chuang-Chuang Li, Southern University of Science and Technology and Shenzhen Bay Laboratory, both Shenzhen, China, and colleagues have performed the first total syntheses of bufogargarizins A and B. The team started from commercially available sitolactone, which was transformed into a vinyl ether cyclopropane-yne intermediate. This intermediate then underwent a key step, i.e., an intramolecular Ru-catalyzed [5 + 2] cycloaddition reaction to obtain the desired [7–5–6–5] tetracyclic core. According to the researchers, this reaction is the first example of a transition-metal-catalyzed [5 + 2] cycloaddition reaction of a vinyl ether cyclopropane-yne
After the tetracyclic core was constructed, several functional group interconversions gave bufogargarizin A. The team also performed a synthesis of bufogargarizin B, in which the [5–7–6–5] core of bufogargarizin B was diastereoselectively reassembled from a [7–5–6–5] tetracyclic skeleton via retro-aldol/transannular aldol cascade reactions. Bufogargarizins A and B were obtained in 28 and 30 steps, respectively, from sitolactone. The strategies used in this work could also enable syntheses of other bufogargarizins and their analogues.
- Asymmetric Total Synthesis of Twin Bufogargarizins A and B,
Li-Ping Zhong, Rui Feng, Jing-Jing Wang, Chuang-Chuang Li,
J. Am. Chem. Soc. 2023.
https://doi.org/10.1021/jacs.2c13494