Vinyl-substituted alcohols are useful intermediates for further transformations in organic synthesis. They can be prepared, e.g., via an alkenylation of aldehydes using vinyl metal reagents or an allylic substitution with oxygen-based nucleophiles. However, these approaches have drawbacks such as a need for sensitive reagents, harsh conditions, or pre-synthesized precursors. Thus, reactions that give vinyl-substituted alcohols from abundant chemical feedstocks under mild conditions would be useful.
Shifa Zhu, South China University of Technology, Guangzhou, China, and colleagues have developed a nickel-catalyzed synthesis of vinyl-substituted alcohols under mild conditions that uses abundant acetylene as a C2 building block. The team reacted different aryl or alkyl aldehydes with acetylene and triethyl silane (SiEt3H), using Ni(cod)2 (cod = 1,5-cyclooctadiene) as a catalyst together with an N-heterocyclic carbene (NHC) ligand precursor and KOtBu as a base. The reactions were performed in tetrahydrofuran (THF) at 35 °C.
The desired silyl-protected vinyl-substituted alcohols (general structure pictured) were obtained in mostly moderate to high yields. They can easily be deprotected and used in further transformations. The reaction has a broad substrate scope and can be performed on a gram scale. The team proposes a reaction mechanism that involves a five-membered oxa-metallacycle intermediate, which is created by a cyclometallation of the aldehyde and the acetylene. A silane-mediated σ-bond metathesis and a reductive elimination then give the desired product.
- Synthesis of Vinyl-Substituted Alcohols Using Acetylene as C2 Building Block,
Zhicong Lin, Boxiang Liu, Yu Wang, Siju Li, Shifa Zhu,
Chem. Sci. 2023.
https://doi.org/10.1039/D2SC06400F
Update (January 5, 2023)
The text originally used “vinyl alcohol” in place of “vinyl-substituted alcohol”. This has been corrected.
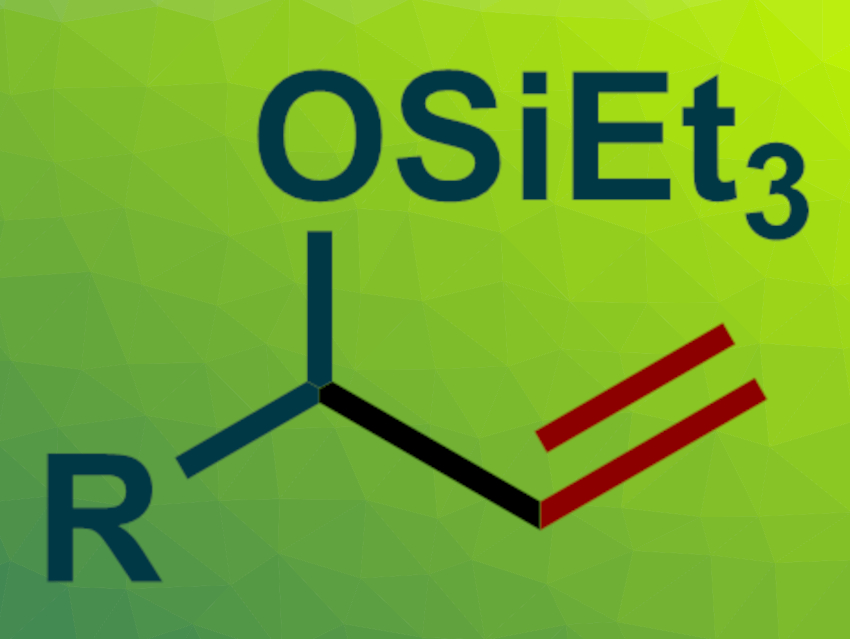




The general structure pictured is incorrect, it represents an silyl protected allylic not a silyl protected vinylic alcohol
Thank you very much for pointing this out! We have changed the text to better reflect the synthesized structure in accordance with the underlying research paper.