In organic synthesis, the core carbon skeleton of a target compound is often prepared first, followed by transformations at the periphery of the molecule and functionalization reactions. “Editing” the skeleton of a compound in later stages could be useful. Skeletal editing could be helpful, for example, in drug discovery to tune the biological properties of a drug candidate.
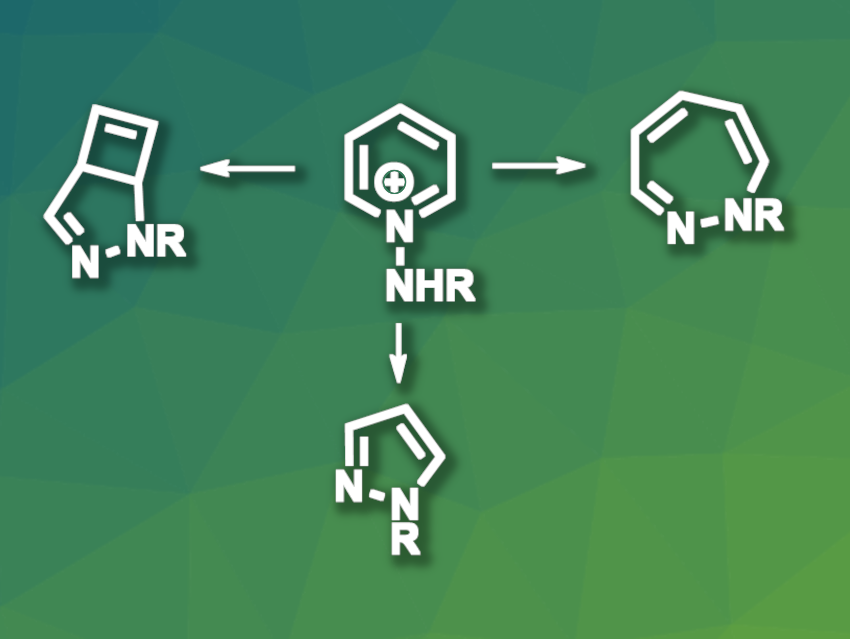
K. N. Houk, University of California, Los Angeles, USA, Ke Zheng, Sichuan University, Chengdu, China, and colleagues have developed a one-pot photochemical skeletal editing protocol for the transformation of pyridines into bicyclic pyrazolines, pyrazoles, or 1,2-diazepines. The approach allows for the selective removal of specific carbon atoms from the pyridine skeleton, can be used in flow chemistry systems, and enables the late-stage modification of drug-like compounds. The team used different pyridinium salts (cation pictured, R = Bz) and irradiated them with LED light, first at 365 nm and then at 420 nm, in dichloromethane at room temperature under a nitrogen atmosphere.
The researchers observed the formation of bicyclic pyrazoline products (pictured above on the left) in moderate to high yields. The reaction proceeds via ring-expanded 1,2-diazepine intermediates (pictured above on the right), which could be isolated when only the 365 nm LED light was used. When the bicyclic pyrazolines were again subjected to 365 nm light in the presence of xanthone as a catalyst, they underwent selective two-carbon deletions to give pyrazoles (pictured above in the bottom row). The 1,2-diazepines could also be converted to pyrazoles by selectively “deleting” specific carbons in the presence of toluenesulfonic acid (TsOH), complementing the selectivity of the deletion via bicyclic pyrazolines.
- Photochemical Skeletal Editing of Pyridines to Bicyclic Pyrazolines and Pyrazoles,
Jiajing Luo, Qingyang Zhou, Zhou Xu, K. N. Houk, Ke Zheng,
J. Am. Chem. Soc. 2024.
https://doi.org/10.1021/jacs.4c03713


![A Path to Substituted Bicyclo[2.1.1]hexanones](https://www.chemistryviews.org/wp-content/uploads/2024/10/1substitutedbicyclo211hexan2ones_2024-125x94.png)
