Every day until December 24, we will unlock a new destination for your bucket list.
In the process, we zigzag through Europe. We start in the southwest and end in the southeast. Of course, you can also visit these destinations in any other order or in individual tours, perhaps combined with other sights—the important thing, we think, is that you do not miss these highlights. For now, have fun discovering!
The start of an exciting chemistry trip through Europe:

1 Chemistry Lab and Teaching Auditorium from the 18th Century
Coimbra, Portugal
See the destination
Let yourself be transported to the academic world of the 18th century. In Coimbra, there is an outstanding chemistry laboratory that has been preserved from the 18th century. Today it is integrated into the Science Museum of the University of Coimbra (MCUC) along with a teaching room that has also been preserved. It is one of the very few original classrooms and laboratories. In addition to many historic exhibits, there is also a video about the laboratory shown in the museum.
The University of Coimbra, founded in 1290 by King D. Dinis, is one of the oldest universities in the world. In 2013, UNESCO declared the university a World Heritage Site, noting its architecture, unique culture and traditions, and historical role.
The Chemistry Laboratory is the most important neoclassical building in Portugal. It was built in the 18th century to teach experimental chemistry during the university reform initiated by the Marquis of Pombal. It embodies the Enlightenment ideal of hands-on science education. Since 2006, the building houses part of the scientific collections of the Science Museum.
Vera Koester
- Chimico Laboratory, Science Museum, Largo Marquês de Pombal, Coimbra, Portugal
Onwards to Spain …

2 Thought-Provoking Periodic Table
Cáceres, Spain
See the destination
What is the best way to represent the periodic table of the elements? A question that keeps chemists busy [1]—but not only them. In 2016, artist Cristina Lucas chose a representation that probably wouldn’t have occurred to us chemists, but which I think is great: The elements are sorted according to their value (€/kg) on the stock market. A huge vertical table shows in real time how the value of each of the elements of the periodic table fluctuates.
Cristina Lucas was born in Jaén, Spain, in 1973 and lives and works in Madrid, Spain. Her work is very diverse and reflects her curiosity and questioning of her surroundings and the world. Again and again her work is also inspired by natural sciences. For example, she developed painting materials and abstract compositions for a series of paintings made from trace elements and minerals that are part of the human body.
Elemental Order is on display at the Museo Helga de Alvear, Cáceres, Spain. The museum grew out of the private collection of Helga de Alvear, the German wife of a Spanish architect. The collection now includes more than 3,000 works. The Museum of Modern Art, opened in 2010, was expanded with a new building in 2021. The collection includes works by more than 500 different artists, including Joseph Beuys, Dan Flavin, Nan Goldin, Paul Klee, and Roy Lichtenstein.
Vera Köster
- Elemental Order, computer program, LED screen, 2016, Cristina Lucas
- Museo Helga de Alvear, Cáceres, Spain
Also of Interest
[1] David Bradley, Periodic Debate, ChemistryViews 2011. https://doi.org/10.1002/chemv.201000093
We continue our travel—on to Paris …

Photo: © Jérémy Mathur/Musée Curie
3 World-Changing Research in a Small Laboratory in France
Paris, France
See the destination
Marie Curie, born as Maria Skłodowska in Warsaw, Poland, is a role model and hero to many female chemists, in particular. With her groundbreaking work on radioactivity and the discovery of the elements polonium and radium, honored by two Nobel Prizes (for Physics in 1903 and for Chemistry in 1911), she was clearly a trailblazer. Curie is also famous among the less scientifically inclined—so you might just get all your family members to come along for a visit to the Musée Curie in Paris, France!
Marie Curie and her family—her husband Pierre Curie, her daughter Irène Joliot-Curie, and her son-in-law Frédéric Joliot-Curie also won Nobel Prizes—are the focus of the museum. Marie Curie spent most of her life and career in Paris, she conducted some of her most important work on radioactive elements in a converted shed because her laboratory lacked the necessary space. The museum is situated in the former Radium Institute, which was founded in 1909 and included a physics and chemistry laboratory which were directed by Marie Curie.
The small museum retraces the different stages of the discoveries of the Curies, “the family with five Nobel prizes”. It shows how radium was used for its “healing properties” in health and beauty products before mankind was aware of its dangers. Visitors can “travel back in time” and see a small chemistry laboratory that was decontaminated and rebuilt in 1981, and some of the original instruments, devices, and glassware preserved from Curie’s time are on display.
The museum also dedicates a part of its exhibition to the Curie Foundation, which was established in 1920 and aimed to develop radiotherapy treatments against cancer. Marie Curie herself tragically died of leukemia in 1934, probably caused by her long-term exposure to radiation during her work. Today, the successor of the Curie Foundation, the Institut Curie, is a leading medical, biological, and biophysical research center and still specializes in cancer research and treatment.
Catharina Goedecke
- Musée Curie, Paris, France
- Facebook page of the museum
Just a short trip by ferry, train, or tunnel brings us to London next …

4 So Much to See and Do for Scientists of all Ages!
London, UK
See the destination
The Science Museum in London, UK, really is a must-see for anyone interested in science, even kids! It shows a vast collection of historic objects that chart 250 years of science and technology—my personal highlights are a DNA molecular model built by Francis Crick and James Watson (pictured), penicillin mould from Fleming’s laboratory, and some of the earliest computers ever made.
The museum also features the Wellcome Galleries, which show some of the most significant medical collections in the world. There, you can find art focusing on the topic, anatomical models, and objects and medical instruments from leech jars to the world’s first MRI scanner. There also is an entire Victorian pharmacy!
However, you do not just have to stare at history in awe: The museum has great interactive galleries, where, for example, children can learn about the patterns all around us, teenagers can test their engineer-like problem-solving skills and explore different technical careers, and everybody can see stories about climate science and the transition to renewable energy. There are also special exhibitions on topics such as science fiction or the race for the COVID-19 vaccine.
Entry to the museum’s general exhibition is free (donations are accepted), and free tickets for a particular time slot can be pre-booked online. My personal tip is to reserve some of your souvenir budget for your visit to the Science Museum, because the museum’s shop is a veritable wonderland of science toys, kits, and gadgets as well as themed clothes, home goods, and books.
Catharina Goedecke
- Science Museum, London, UK
While we are in the UK, let us follow in the footsteps of Watson and Crick to Cambridge …

5 The Blueprint of Life and a Nice Cold Pint
Cambridge, UK
See the destination
Cambridge is home to one of the oldest and most famous universities in the UK. While there have been many important discoveries made here, one of the most famous is the discovery of the structure of DNA by Francis Crick and James Watson at the University’s Cavendish Laboratory in 1953 [1].
Close to the Cavendish Laboratory, there was “The Eagle”, a pub that was a popular haunt for researchers, staff, and students of the university. Famously, on February 28, 1953, Crick came to The Eagle around lunchtime and announced to the pub’s guests that he and Watson had discovered the double helix structure of DNA—a breakthrough that provided important insight into how genetic information is stored and copied. The pub (now one of the oldest in Cambridge) is still there, and a plaque outside (pictured) commemorates this huge moment in scientific history. Inside, you can enjoy a pint of “Eagle’s DNA” beer.
Francis Crick and James Watson, together with Maurice Wilkins, received the 1962 Nobel Prize for Physiology or Medicine for their discovery. Since a good controversy is always interesting: Watson’s and Crick’s discovery was based on X-ray diffraction results of DNA, which had been produced by Rosalind Franklin and her graduate student Raymond Gosling. There has been some debate over the contribution of Franklin to the discovery and if she should have received more credit [2].
While you’re in Cambridge, you can also visit the university’s museums and botanic garden or go on one of many walking tours through the city and see the university’s famous colleges.
Catharina Goedecke
- The Eagle Pub, Cambridge, UK
- University of Cambridge’s museums
References
[1] 100th Birthday: Francis Crick, ChemistryViews 2016.
[2] Catharina Goedecke, 100th Birthday: Rosalind Franklin, ChemistryViews 2020. https://doi.org/10.1002/chemv.202000065
Continuing high into the north …

6 World’s Largest Iron Ore Mine
Kiruna, Sweden
See the destination
The iron ore mine in Kiruna is said to be the largest and most modern iron ore mine in the world. The huge magnetite deposit has been mined for around 120 years. The mine is owned by LKAB (Luossavaara-Kiirunavaara Aktiebolag), a large Swedish mining company that extracts high-grade phosphorus-bearing magnetite from it.
Kiirunavaara was first mentioned in writing in 1696, but for a long time, ore mining was not worthwhile despite the rich deposit. The site was too remote in northern Sweden in an inhospitable environment, and the phosphorus iron ore could not be processed with the technologies available at the time. The ore deposit only became interesting with the invention of the Thomas process in the 1870s, which made it possible to process ores containing phosphorus. After the completion of the Swedish ore railroad, mining on an industrial scale began in 1900.
Visitors are taken by bus into the mountain to an underground visitor center. This show mine extends over 20,000 m² and illustrates ore mining and iron production in an authentic mining environment with display boards, films, and machines. A mining museum tells the centuries-long history of the mine and the changing technology of iron ore mining.
The millions of tons of iron ore mined in Kiruna each year are still transported to Narvik in Norway via the more than 470 km long railroad line. There is an ice-free harbor there, through which the raw material processed into granules can be transported away by ship all year round.
Vera Koester
- LKAB:s Guided Tours
- LKAB’s Visitor Center
- LKAB‘s website – Europes largest iron ore producer
On to another mining-based destination …

Photo of plaque: Uwezi, wikimedia commons, CC BY-SA 4.0
7 The Mine that Gave Rise to Many New Elements
Ytterby, Sweden
See the destination
The Ytterby mine, or Ytterby gruva in Swedish, is situated about a half-hour’s drive northeast of Stockholm on the island of Resarö. It might not look very exciting at first glance, but for chemists (and the periodic table), it is a very important place: In this small historic quartz and feldspar mine, minerals were found that led to the discovery of eight new elements. This makes the small village of Ytterby the most important place to the periodic table.
Four of these elements are named after Ytterby, i.e., yttrium (Y), ytterbium (Yb), terbium (Tb), and erbium (Er). The others are scandium (Sc), named after the Latin scandia meaning “Scandinavia”, holmium (Ho), named after the Latin Holmia for Stockholm, thulium (Tm), named after Thule, a northerly location mentioned in ancient Greek and Roman literature, and gadolinium (Gd), named after the chemist Johan Gadolin [1].
History
Ytterby gruva opened in the 1600s to extract quartz for iron production. In the 1700s and 1800s, Ytterby was mined for feldspar, which was used by the glass and porcelain industries. In 1787, Carl Axel Arrhenius visited the Ytterby mine and its elemental history began.
Arrhenius was an officer in the Swedish army and had an interest in geology and chemistry. He found an unusually heavy black mineral there. A sample of the mineral was sent to Johan Gadolin, who performed a chemical analysis and concluded that up to 38 % of the stone consisted of previously unknown elements, or an unknown “earth”. The mineral that Arrhenius found was later named gadolinite (chemical composition Y2FeBe2Si2O10), and served as the source of several previously undiscovered rare earth elements.
Rare earths intermingle almost indistinguishably, and where you discover one, you find many others. Sweden has rich deposits of rare earths. In addition, during the last ice age, the glaciers eroded the uppermost soil layers in large parts of Sweden, so that these deposits can be easily mined.
Today
ASM International, formerly known as the American Society for Metals, designated the mine as a historical landmark in 1989, which has been commemorated with a plaque at the site (see photo) [2]. In 2015, the association Ytterby Gruva was founded, which aims to spread and preserve knowledge about the mine and its history. In 2019, a EuChemS Historical Landmarks Award plaque was unveiled at the entrance of Ytterby mine [3].
Visitors can explore the area around the mine entrance on their own. The mine itself cannot be visited, but since it is designated as a natural monument, visitors may not take rock samples with them. Some streets in the area are named after elements.
Catharina Goedecke
- Michelle Maccrackin, Ytterby elements: field trip, Baltic Sea Center, Stockholm, Sweden 2017. (accessed November 29, 2022)
References
[1] 165th Anniversary: Death of Johan Gadolin, ChemistryViews 2017.
[2] ASM Historical Landmarks, www.asminternational.org. (accessed November 28, 2022)
[3] EuChemS Historical Landmarks Award plaque to be unveiled at Ytterby mine, www.euchems.eu. (accessed November 28, 2022)
From Ytterby, it is just a short trip to Stockholm …

8 Nobel Prize Museum
Stockholm, Sweden
See this destination
Opened in 2001, the museum displays information about the Nobel Prize, Nobel Prize Laureates from 1901 to the present day, and the life of Nobel Prize founder Alfred Nobel (1833 – 1896). It is housed together with the Swedish Academy and its Nobel Library in the former Stockholm Stock Exchange building in the center of the old town. After completion of the planned Nobel Center—a house for science, culture, and dialogue to be located at Slussen in central Stockholm, the museum is to move there. The start of construction is planned for 2026, with a two-year construction period. Inauguration may occur during 2029 [1].
The Nobel Prize Museum wants to encourage reflection on how we can make the most of our creativity and what significance our environment can have for the creative process. Objects and films illustrate the achievements of different Nobel Laureates. Among them, of course, many chemists.
It is a small museum and many of the exhibits are donated. Nobel Laureates who visit the museum donate items such as a book or tie and sign a chair in the museum’s bistro. Many of these items are not necessarily directly related to the field of expertise of the Nobel Laureate.
There were also spectacular donations: A manuscript containing a variant of Einstein’s 1915 general theory of relativity with comments by Nobel Laureate Max von Laue. It was the basis for a paper published by the Royal Prussian Academy of Sciences in December 1922. Until 1948, the manuscript was in the possession of Max von Laue. After 1948, it had various private owners. In the fall of 2018, it was acquired by Åsa and Per Taube of Stockholm to donate it to the collection of artifacts housed at the Nobel Prize Museum. The manuscript has been on public display since the end of 2019.
In addition, the museum offers touch screens and printed posters with general information about the prizes and Laureates. A large part of the museum is used for temporary exhibitions.
Vera Koester
- Nobel Prize Museum, Stockholm, Sweden
Admission is free on Tuesday evenings
Reference
[1] New Nobel Center takes the next step, Press release from the Nobel Foundation, May 4, 2022. (accessed November 23, 2022)
Keeping on the path of Alfred Nobel …

9 Remains of Alfred Nobel’s Blasting Experiments
Stockholm, Sweden
See the destination
In 1867, Alfred Nobel (1833 – 1896) patented dynamite, which made him world famous. It all began not entirely happily in Vinterviken in the south of Stockholm. Even today, you can see some remains of Alfred Nobel’s blasting experiments there. These are mainly tunnel-like holes in the hill.
In 1865, Alfred Nobel bought a farm in Vinterviken to produce nitroglycerine. With the construction of several railroad tunnels in the area, demand for the explosive was high. The site in Vinterviken was surrounded by hills so that the adjacent areas were protected from accidental explosions, but it was still close to Stockholm and on the water, which made it possible to build quays.
Initially, the facilities were simple sheds, and some production took place outdoors. In the spring of 1866, there were several serious accidents in which many employees lost their lives. Alfred Nobel had already lost his brother in a similar accident a few years earlier. So he started experimenting with different explosives and absorbents to make the explosive safer to handle and transport. By accident, at one point he mixed nitroglycerin with diatomaceous earth and obtained a soft, pliable, dough-like material that was relatively stable, and easy and safe to handle. He succeeded in detonating the material with his blasting cap.
First, Alfred Nobel sold dynamite as “Nobel’s Blasting Powder”. Later he decided to change the name to dynamite, from the Ancient Greek word dýnamis (δύναμις), meaning “power”.
The former explosives factory at Vinterviken today houses a restaurant. A few blasting bunkers can still be found along the waterfront. From Aspudden metro station (which is less than 15 minutes from the main train station), it is about a 15-minute walk to Vinterviken. The tunnels are signposted at the Stockholm City Museum, and the coordinates are N 59.3106 E 17.9863.
Vera Koester
Reference
[1] Jozef Brodala, The Farm That Helped Birth the Genius of Alfred Nobel, The Culture Trip December 22, 2017. (accessed November 23, 2022)
Next, we travel back south to the Netherlands …

Photo: Yann Caradec, wikimedia commons, CC BY-SA 2.0
10 The Small Organisms that Shape Our World
Amsterdam, The Netherlands
See the destination
Micropia is a museum in Amsterdam, The Netherlands, that focuses on microorganisms. This “microbe” zoo shows visitors the importance of the smallest organisms for our daily life in an inspiring and interactive way. It is situated in the historical building “Ledenlokalen” in central Amsterdam, next to the ARTIS zoo, which houses significantly larger organisms, such as elephants or giraffes.
Micropia aims to show a usually invisible world and introduce a wide public to microorganisms. Visitors can look into microscopes and Petri dishes and see bacteria, algae, fungi, viruses, and tiny organisms such as tardigrades (also known as water bears). Microbes living in and on the human body are shown and explained, such as the mites that live on eyelashes. A “body scanner” shows visitors what types of microbes live on their body, and a “kiss-o-meter” counts the number of microbes transferred during a kiss.
The museum shows how important microorganisms are for life on Earth, from oxygen production to the recycling of organic materials, fermentation, and the importance of intestinal bacteria for digestion. It explains how some microbes live in the most extreme places such as geysers, the deep sea, or nuclear reactors, but also shows what can “lurk” on everyday household objects.
During your visit, you can collect your favorite microbes using a stamp card. Activities such as “Lab Talk” explain, e.g., how fungi recycle materials. The museum also offers an education program that allows students to discover the world of microbes and provides them with an idea of possible careers in microbiology.
The organization ARTIS was founded under the name Natura Artis Magistra in 1838 with the objective of “promoting the knowledge of natural history” and has transitioned from running the zoo to a broader educational institution. In addition to the zoo, a planetarium, and Micropia, it also operates the Groote Museum, where visitors experience the connection between everything that lives and explore the “big questions” of life. Combined tickets are available.
Catharina Goedecke
- Micropia, Amsterdam, The Netherlands
Open seven days a week. Tickets are EUR 17.50. For children under 13 years of age, admission is free.
Also of Interest
Speaking with a Microbe: Interview with Fridolin E. Coli
Fridolin E. Coli, gram-negative rod-shaped bacterium, talks to ChemistryViews about his more than one hundred years of experience in research
Speaking with a Microbe: Interview with James T. Aquaticus
James Thermus Aquaticus, a Gram-negative thermophilic bacterium, talks about his involvement in the most important enzymes in molecular biology
On to Brussels …

11 Iron Crystals at a World’s Fair
Brussels, Belgium
See the destination
The Atomium is a well-known landmark and a must-see in Brussels, Belgium, but not every visitor might know where its shape comes from: It is a representation of the body-centered cubic unit cell of an iron crystal, magnified 165 billion times. It was planned as the flagship building of the 1958 Brussels World’s Fair, or Expo 58, as a symbol of scientific progress and the peaceful applications of atomic research after the use of nuclear weapons in World War II.
The Atomium project was led by the engineer André Waterkeyn, who was Head of the economic department of Fabrimétal, an association of companies in the metal-fabricating industry. Waterkeyn was joined by the architects André and Jean Polak. Construction began in March of 1956 and the Atomium was completed in March of 1958, just in time for the World’s Fair. The framework was built using steel, and the outer shell was made from aluminum. The shell of the spheres is composed of triangular panels, and windows in some of the spheres allowed visitors to enjoy the view over the Expo. An exhibition highlighting the peaceful uses of nuclear energy was installed in four spheres, and a restaurant was created in the top sphere. Visitors moved between the spheres through tubes that connect the “atoms”.
Originally, the Atomium was not intended to survive long after the World’s Fair in 1958. The city of Brussels took over operations and kept it open to visitors. Maintenance was lacking, and the Atomium became somewhat run-down. It was almost demolished in the 1990s due to a lack of visitors and funds. However, it also became a landmark and a symbol of Brussels, and a major renovation project took place from 2004 to 2006. The outer aluminum shell was removed completely, and the metal was sold to help finance a new metal shell. This new shell was made from stainless steel, which increased the weight of the structure by 100 tonnes. A visitor’s center was built at the base.
Today, the Atomium’s spheres house a permanent exhibition that retraces the history of the building and of Expo 58 with photos, videos, and documents from the era as well as detailed models of the fair. There are also changing temporary exhibitions, a restaurant in the highest sphere, and amazing views from the windows that are a draw for photographers.
Catharina Goedecke
- Atomium, Brussels, Belgium
The Atomium is open for visitors seven days a week, with tickets costing EUR 16 for adults, EUR 8.50 for children up to 17 years of age, and EUR 14 for seniors. Admission for children shorter than 1.15 m is free. Guided tours for groups are available in several European languages.
After very large “particles”, on to much smaller ones …

Photos: © CERN
12 The Largest Laboratory for the Smallest Particles
Geneva, Switzerland
See the destination
While chemists are usually mostly concerned with electrons and atomic nuclei, there is a veritable “zoo” of subatomic particles for researchers to discover. The largest particle physics laboratory in the world is CERN (short for Conseil Européen pour la Recherche Nucléaire or European Organization for Nuclear Research) in Geneva, Switzerland, right on the border with France.
The convention establishing CERN as a joint laboratory was signed in 1953 by twelve European countries and entered into force in 1954. Originally, CERN was meant to study nuclear physics, but the focus soon shifted to particle physics.
Research
CERN hosts particle accelerators such as the well-known Large Hadron Collider (LHC, pictured above on the left), which is the world’s largest and most powerful particle accelerator and was built between 1998 and 2008. The LHC is situated in a tunnel with a circumference of 27 km beneath the Swiss–French border. Like other colliders, it is used to collide beams of particles with very high kinetic energies and then detect the products formed by the collision. There also are computing facilities and a major network hub to handle all the data generated at the site.
Key achievements made at CERN include the discovery of W and Z bosons in 1983 [2], the first creation of antihydrogen atoms in 1995 [3], and the discovery of a boson consistent with the long-sought Higgs boson (or “God particle”) in 2012 [4,5]. Besides such breakthroughs, research at CERN also benefits society somewhat more directly by creating new technology and expertise that can be applied to fields beyond high-energy physics, e.g., in materials science or computing. For example, CERN is considered the birthplace of the World Wide Web [1]. Particle accelarators also serve purposes apart from basic research, e.g., in medical diagnosis and therapy.
Visit
Visitors are welcome at CERN to see exhibitions or participate in guided tours or public events. The Globe of Science and Innovation (pictured above on the right) hosts an exhibition for visitors called Universe of Particles. It explains where the particles in the universe come from, what laws govern their behavior, and which big questions of contemporary physics are currently being explored by CERN. It also features the first web server and a video show about the origins of the universe (in French).
CERN visits are free of charge. The “Science Gateway”, CERN’s new visitor center and a flagship project for science education and outreach, will open in 2023. It will host new exhibitions, labs for hands-on experiments, a large auditorium, a shop, and a restaurant.
Catharina Goedecke
References
[1] J. Gillies, R. Cailliau, How the Web was Born: The Story of the World Wide Web. Oxford University Press, 2000. ISBN: 978-0-19-286207-5.
[2] R. Cashmore, L. Maiani, J.-P. Revol, Prestigious Discoveries at CERN. Springer Berlin Heidelberg, 2003. ISBN: 978-3-642-05855-4.
[3] G. Baur at al, Production of antihydrogen, Phys. Lett. B. 1996, 368, 251–258. https://doi.org/10.1016/0370-2693(96)00005-6
[4] G. Aad et al., Observation of a new particle in the search for the Standard Model Higgs boson with the ATLAS detector at the LHC, Phys. Lett. B. 2012, 716, 1–29. https://doi.org/10.1016/j.physletb.2012.08.020
[5] S. Chatrchyan et al., Observation of a new boson at a mass of 125 GeV with the CMS experiment at the LHC, Phys. Lett. B. 2012, 716, 30–61. https://doi.org/10.1016/j.physletb.2012.08.021
Continuing in Switzerland …

13 Great View Over Zurich and Start of ETH’s Chemistry Department
Zurich, Switzerland
See the destination
The main building of the ETH Zurich was built in 1864 according to plans by Gottfried Semper. The observation terrace in front of the main building is called Polyterrasse. From here you have a fantastic view over the old part of Zurich. At the edge of the terrace is a cafeteria of the university with an outside seating area, where you should definitely have a coffee or beer when the weather is nice. Here, you can immerse yourself in the early days of ETH, which enjoys worldwide a high reputation in science and industry, and plan the rest of your visit.
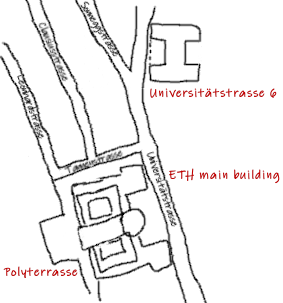
The Beginnings of the Renowned ETH Chemistry Faculty
The ETH started in 1855 with six faculties with 231 students and 30 professors. The faculties, which were at various sites throughout the city of Zurich, were architecture, civil engineering, mechanical engineering, chemistry, forestry, and an integrated department for mathematics, natural sciences, literature, and social and political sciences. Initially, there was a chemistry building in front of the main building, built between 1859 and 1861, but it was very quickly replaced by a new building.

The new building (pictured above) was constructed between 1884 and 1886 by the architects F. Bluntschli and G. Lasius according to a program of two chemistry professors, V. Meyer and G. Lunge. The size of the chemistry department increased from 1700 m² to 8000 m², and the interior was very modern and pioneering for the time: There were large, bright teaching and working rooms and good conditions for experiments in the lecture hall as well as for work in the laboratory. You can visit the building from the ourside; it is in a few minutes walk from ETH’s main building.
The chemistry building housed many first-class chemists over 100 years, including Richard Willstätter (1872–1943), Herman Staudinger (1881–1965), Leopold Ruzicka (1887–1976), Vladimir Prelog (1906–1998), and Richard Ernst (1933–2021), many of whom were awarded the Nobel Prize for their research in this building and contributed to the outstanding reputation of chemistry at ETH Zurich today.
The Chemistry Faculty Today
The building was extended between 1955 and 1974 and expanded to an area of 24,000 m². However, it eventually reached its maximum capacity and became too small to accommodate the rapidly growing field of chemistry. In November 2001, a modern chemistry building with 35,000 m² opened on the Science City campus on the Hönggerberg in a nice park (pictured below).

On December 9, 2010, the former chemistry building at Universitätstrasse 6 was honored as a “Historic Site of Chemistry” and, thus, as the place of work and education of several generations of chemists and chemical engineers. Since 1987, it is listed as a historical monument.
Vera Koester
- Historic Site of Chemistry Building, Universitätstrasse 6, Zurich, Switzerland
- bQm Kulturcafe & Bar, ETH Zentrum, Polyterrasse, Leonhardstrasse 34, Zurich, Switzerland
References
Barbara Winter-Werner, Detlef Günther, Chemical Landmark 2010 – Designation of the Old Chemistry Building of ETH Zurich, Chimia 2011, 65(6), 447-448. https://doi.org/10.2533/chimia.2011.447
Chemical Landmark 2010, The Old Chemistry Building of ETH Zurich, akademie der naturwissenschaften (scnat), Bern, Switzerland. (accessed November 21, 2022)
Update December 15, 2022
The article first showed a photo of the Irchel campus of the University of Zurich instead of a photo of ETH Hönggerberg.
On to Germany for some “elemental” research …

Fotos: Experiment setup in the heavy ion storage ring (ESR) and ion trap SHIPTRAP. © J. Hosan/GSI Helmholtzzentrum für Schwerionenforschung GmbH
14 Birthplace of Six Superheavy Elements—A Look Into the Research Facility
Darmstadt, Germany
See the destination
The discovery of elements has changed extremely over the years. Today, extremely sophisticated equipment and complex theories are needed—and you need a lot of space. Money, too, of course. Are you interested in seeing how something like this works? Or how to do experiments with elements that exist only for very, very short moments?
GSI
Between 1981 and 1996, six elements with atomic numbers from 107 to 112 were discovered and produced at the facilities of the GSI Helmholtz Center for Heavy Ion Research in Darmstadt, Germany: Bohrium, Hassium, Meitnerium, Darmstadtium, Roentgenium, Copernicium. The GSI operates a worldwide leading accelerator facility for research purposes.
Superheavy elements (SHE) are elements with proton numbers (Z) of 104 or greater. They are produced by fusing atomic nuclei of lighter elements using particle accelerators. The GSI used this technique in the discovery experiments for the above-mentioned elements by bombarding elements 82 (lead) and 83 (bismuth) with moderately heavy projectile nuclei (Z = 24, 26, 28, and 30).
FAIR—The Universe in the Lab
The international particle accelerator facility FAIR (Facility for Antiproton and Ion Research in Europe) is currently being built in Darmstadt. This is currently one of the largest and most complex construction sites worldwide and will become one of the largest research projects worldwide. In FAIR, matter that otherwise only exists in the depths of space will be created. FAIR will generate particle beams of unprecedented intensity and quality. Ions of all natural elements of the periodic table, as well as antiprotons, will be accelerated. The particles will be accelerated to almost the speed of light. Scientists from all over the world will be able to gain new insights into the structure of matter and the evolution of the universe from the Big Bang to the present.
The core of FAIR is a superconducting ring accelerator with a circumference of 1,100 m. Connected to it is a complex system of storage rings and experimental stations. The existing GSI accelerators will serve as the first acceleration stage.

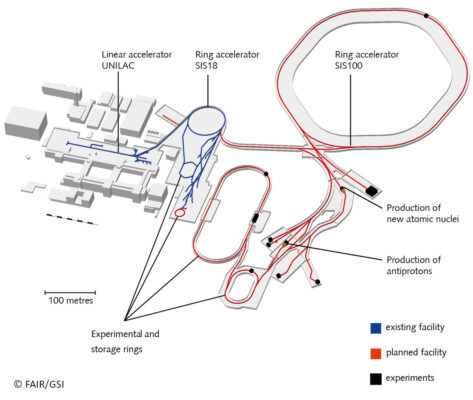
Future ring accelerator SIS100, September 2020. Fotos: © D. Fehrenz/GSI/FAIR
Green IT Cube
The Green IT Cube, a high-performance computer center, is currently being built to analyze the huge amounts of data from the experiments. When completed, it will have a cooling capacity of twelve megawatts, making it one of the largest scientific data centers in the world.

Supercomputer L-CSC. Foto: © Thomas Ernsting, HA Hessen Agentur GmbH
Visits
In addition to their research, the GSI offers a few slots where visitors can get an introduction to their work. You can see their huge, fascinating facilities. For the layman, they look a bit like space research. The visit begins with an introductory lecture, then selected research stations will be visited. It is also possible to visit the construction site of the FAIR particle accelerator facility. Altogether it takes about two hours.
GSI is a publicly funded research institution, therefore, a visit is free of charge. More details can be found on the webpage: https://www.gsi.de/en/media-news/guided_tours
Vera Koester
- GSI Helmholtzzentrum für Schwerionenforschung
FAIR – Facility for Antiproton and Ion Research
Planckstraße 1, 64291 Darmstadt, Germany
Also of Interest
Vera Koester, Michael Block, Studying Superheavy Elements, ChemistryViews 2019. https://doi.org/10.1002/chemv.201900122
Vera Koester, Christoph E. Düllmann, The Chemistry of Superheavy Elements, ChemistryViews 2019. https://doi.org/10.1002/chemv.201900123
Klaus Roth, New Kids on the Table: Is Element 118 a Noble Gas?, ChemistryViews 2018. https://doi.org/10.1002/chemv.201800029
From Darmstadt, it’s only a short trip to Gießen …

15 Justus Liebig’s Laboratory—From Alchemy to Founder of Organic Chemistry
Gießen, Germany
See the destination
The historic laboratory of Justus von Liebig, where he worked from 1824 to 1852, has been a museum since 1920, making it one of the oldest surviving laboratory buildings where visitors can experience the working conditions of the time. The Liebig Museum includes a rich collection of historical chemical apparatus, scientific instruments, documents such as chemistry books, letters, pictures, and personal memorabilia, and well-preserved technical equipment such as furnaces, fume hoods, and drains. One can trace the development of laboratories and marvel at how close to the labs we know today Liebig’s latest lab was already.
Justus von Liebig is considered one of the main founders of organic chemistry and is known for his important contributions to agricultural and biological chemistry. Liebig is also considered the founder of chemistry education, combining research and teaching in one laboratory and inspiring students with experimental lectures.
 The museum beautifully shows, among other things, how Liebig founded modern mineral fertilization and the beginning of agrochemistry, developed a manufacturing process for beef extracts, and modern analytical methods. The museum displays, e.g., various versions of his five-ball apparatus (originally called the potash apparatus). Liebig developed it in 1831. Today the logos of the American Chemical Society (ACS) and the younger chemist’s group of Gießen (JungchemikerForum JCF Gießen) carry it in its logo.
The museum beautifully shows, among other things, how Liebig founded modern mineral fertilization and the beginning of agrochemistry, developed a manufacturing process for beef extracts, and modern analytical methods. The museum displays, e.g., various versions of his five-ball apparatus (originally called the potash apparatus). Liebig developed it in 1831. Today the logos of the American Chemical Society (ACS) and the younger chemist’s group of Gießen (JungchemikerForum JCF Gießen) carry it in its logo.
The Justus Liebig Laboratory has been awarded the EuChemS Historical Landmarks as well as the “Historic Sites of Chemistry” (Historische Stätten) of the German Chemical Society (GDCh).
Anyone who wants to relive the path from alchemy to modern chemistry and who wants to be inspired by Liebig and his many ideas and projects should definitely go to Giessen. Since last summer, the young chemists’ group (JungchemikerForum) of the University of Giessen takes over some of the guided tours in the museum.
Vera Koester
- Justus-Liebig-Museum, Liebigstr. 12, Gießen, Germany
Update (December 8, 2022)
Unfortunately, a fire destroyed parts of the historic lecture hall and parts of the analytical, the pharmaceutical laboratory, and the library are damaged mainly by the resulting soot.
Due to the damage, the museum must remain closed until further notice. All events have been canceled. As a consequence, the museum is especially dependent on donations and asks for support: Justus Liebig-Gesellschaft zu Gießen e.V., IBAN: DE66 5139 0000 0003 9916 01 (Volksbank Mittelhessen) or IBAN: DE72 5135 0025 0200 5813 50 (Sparkasse Gießen).
Source: Karola Schepp, Marc Schäfer, Gießener Allgemeine, 06.12.2022, 17:19 Uhr; Rüdiger Schäfer, Gießener Allgemeine, 06.12.2022, 19:20 Uhr
For further information see: https://www.liebig-museum.de/
On to the German capital …

16 Marvel at the Greats of Chemistry
Berlin-Dahlem, Germany
See the destination
In the district of Berlin-Dahlem in Germany, a cosmopolitan city of science and an exclusive residential district were to be created at the beginning of the 20th century. This double ambition, which was filled with life in the course of history, still exists today and invites to an impressive walk.
Many villas built by outstanding architects, small parks, and numerous scientific institutions, including the Free University of Berlin with several institutes, characterize the image of the district today, and one can marvel at the greats of chemistry who have worked here.
A nice circular route runs, for example, from Faradayweg, through Ihmestraße, Garystraße, Boltzmannstraße, Van’t-Hoff-Straße, and Thielallee. Along the way you can see, among many other things,
- the villa of Richard Willstätter (Director of the Chemistry Department of the Kaiser Wilhelm Institute for Chemistry and 1915 Nobel Prize in Chemistry “for his research on plant pigments, especially chlorophyll”) in Faradayweg 10,
- the Kaiser Wilhelm Institute for Cell Physiology (1st Director was Otto Heinrich Warburg who received the 1931 Nobel Prize in Physiology or Medicine for “his discovery of the nature and mode of action of the respiratory enzyme”) in Boltzmannstraße 1. Today it is the archive of the Max Planck Society. Or
- the villa of Fritz Haber and his wife Clara Immerwahr built by the architect Ernst von Ihne in Hittorfstraße 24/Faradayweg 8.
The video below gives more details.
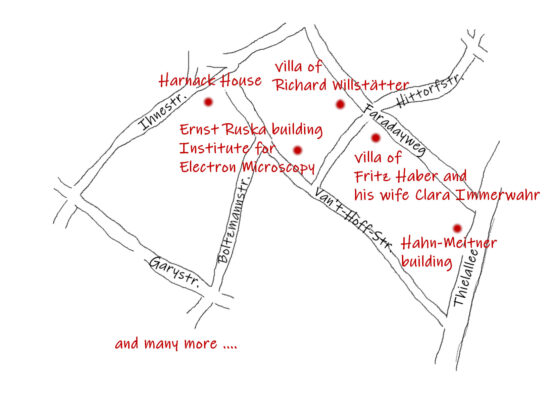
To reform the German science system and prepare it for industrialization, Adolf Harnack, professor of theology in Berlin and close advisor to the emperor (Kaiser in German), proposed that independent research institutes be established alongside the universities to focus on specialized basic research. Following his suggestion, the Kaiser Wilhelm Gesellschaft (KWG; Kaiser Wilhelm Society) was founded in 1911. In 1912, the first institutes of the Kaiser Wilhelm Society moved into their own buildings in Berlin-Dahlem. Each institute was headed by a prominent director such as Peter Debye, Fritz Haber, Otto Hahn, Ernst Otto Beckmann, and Richard Willstätter. Several of them became Nobel Laureates.
At the end of World War II, a new research society, the Max Planck Society, was founded, taking over the properties and staff of the Kaiser Wilhelm Society. The Free University of Berlin (FU Berlin) was founded in Berlin-Dahlem in 1948 and is today located in this area.
Vera Koester
… and further south we go …

Photos: Ostwald’s laboratory and Color Double Cone © Gerda and Klaus Tschira Foundation
17 Insights Into the World of a Polymath
Großbothen, Germany
See the destination
Friedrich Wilhelm Ostwald (born in 1853 in Riga, Livonia, and died in 1932 in Leipzig, Germany) is considered one of the founders of physical chemistry. He received the Nobel Prize in Chemistry in 1909 for his work on catalysis and his studies on equilibrium conditions and reaction rates, and taught at the University of Leipzig. After his retirement, he moved to the “Energy” country estate, a property with five houses in Großbothen near Leipzig, to work as an “independent researcher”, In 1974, Ostwald’s descendants opened the estate to visitors. Today, there is a museum and space for hosting conferences.
In the former music and dining rooms of the Ostwald family, you can today learn a lot about the life of Wilhelm Ostwald. Fascinating is the original preserved library with about 24,000 titles. Oswald had a wide range of interests and also published many books, including several textbooks. In 1887, he founded the Zeitschrift für physikalische Chemie, Stöchiometrie und Verwandtschaftslehre for which he won Jacobus Henricus van ‘t Hoff as co-editor. Ostwald edited the journal until 1922, by which time it had 100 volumes.
Wilhelm Ostwald used his laboratory (pictured above) for numerous experiments, for studies on color theory, and as a painting studio. Today among other things the original laboratory table can be admired in this room. I was very enthusiastic about Oswald’s color theory. The picture shows Wilhelm Ostwald’s Color Double Cone which is on display in the laboratory. Ostwald arranged triangles of 680 standardized color shades with an upper white tip and a lower black tip.
The museum rooms also include depots with scientific equipment, a paint and paper depot with over 1000 painted landscapes and about 3000 color sketches by Wilhelm Ostwald, and a furniture depot. These depots as well as an extensive archive can be used for scientific purposes.
In the sourrounding park, visitors can see a quarry with the Ostwald family’s burial place.
Oswald’s self-sufficient supply system is almost contemporary again: There is a windmill and a horse mill in the park. At the horse mill, the donkeys, August and Lotte took turns running in circles, driving a shaft and several gears so that water was pumped from a well into a large water tank in the attic of the house. This allowed Ostwald to install running water in the house.
On request, the museum offers guided tours with individual focuses such as on chemistry or color theory.
Vera Koester
- Wilhelm Ostwald Park, MUSEUM • TAGUNGEN, Grimmaer Straße 25, Grimma / OT Großbothen, Germany
An institution of the Gerda and Klaus Tschira Foundation
Next stop at the Deutsches Museum …

Photos: New entrance © Alexander Goettert | Deutsches Museum; Lavoisier Laboratory © Deutsches Museum, München | Reinhard Krause Deutsches Museum; München
18 Masterpieces of Science and Technology
Munich, Germany
See the destination
The “Deutsches Museum” (officially Deutsches Museum von Meisterwerken der Naturwissenschaft und Technik or German Museum of Masterpieces of Science and Technology) in Munich, Germany, is one of the world’s largest museums of science and technology. It was founded in 1903 and has five branches, with the main branch on Munich’s “Museumsinsel” in the Isar river in addition to a transport museum in Munich, an aviation museum in Oberschleißheim, and branches in Nuremberg and Bonn that focus on the future and contemporary research, respectively.
The vision of the museum’s founder, Oskar von Miller, was to create a place where science and technology could be experienced in a hands-on way, with experiments and demonstrations. The exhibition building on the Museumsinsel in Munich was opened in 1925.
The museum’s collections include more than 100,000 exhibits, of which about 25,000 are normally on display. The museum also houses Germany’s largest museum library, a leading special archive on the history of science and technology, its own workshops, and a research institute. Usually, the exhibition space on the Museumsinsel is 45,000 m2, but due to extensive renovations, it is currently limited to about 20,000 m2.
The museum features exhibitions on, e.g., mathematics, physics, chemistry, engineering, electronics, energy and motors, optics, photography, health, aviation, agriculture, robotics, and space. The chemistry exhibition shows the importance of chemistry in everyday life. It also shows historical laboratories and has a visitor lab. Interactive elements help visitors understand the concepts and keep children engaged.
Visitors can experience spectacular experimental lectures, trace the development of chemical laboratories since the late Middle Ages, or carry out experiments themselves. One highlight is the “crime scene diorama” where questions such as “What is that white powder on the table?” “Is that blood on the floor?” are answered using chemical analysis.

Photos: © Deutsches Museum, Munich; Reinhard Krause, Deutsches Museum
The exhibition also features, for example, a simple wooden table with the original equipment used to first demonstrate the fission of uranium atoms in 1938 (pictured below on the right), and a periodic table, in which samples of all non-radioactive chemical elements are shown in acrylic glass cubes (pictured below on the left).
Catharina Goedecke
- Deutsches Museum, Munich, Germany
The museum is open seven days a week. A one-day ticket for adults is about EUR 15, with discounts for families, children, students, and groups
Continuing to Italy …

19 Probably The Oldest and Prettiest Pharmacy
Florence, Italy
See the destination
The Officina Profumo-Farmaceutica di Santa Maria Novella is considered the oldest pharmacy in the world. It became known for fragrances, remedies, and herbal products. Today, you can step back in time in the magnificent rooms and museum.
In 1221, Dominican monks came to Florence and founded what would become the city’s first great basilica, the Basilica of Santa Maria Novella. The impressive facade was added in the 15th century by Renaissance architect Leon Battista Alberti. Inside are works of art by some of Italy’s greatest artists, including Giotto, Masaccio, Filippino Lippi, and Ghirlandaio, as well as two cloisters and a museum.
It was common for monasteries to have private gardens where various types of medicinal herbs grew. These were used to make early forms of medicines, balms, and tinctures. Traditionally reserved for the treatment of monks within the monastery, by the 14th-century, news of the healing properties of Santa Maria Novella’s remedies gradually spread outside the monastery walls. Perfumed water was used for a wide variety of medical treatments, including fighting the plague.
The Historic Shop, the main part of the store that can be visited today, was originally built as the Chapel of San Niccolò by the wealthy Florentine merchant Dardano Acciaioli as a thank-you for healing by the Dominican monks. In 1612, the Officina Profumo-Farmaceutica di Santa Maria Novella was officially founded. Orally transmitted recipes and traditions were perfected and preserved, and many of them are still produced today. In the 18th century, the pharmacy was decorated with ornately carved wood paneling, magnificent chandeliers, and frescoes on the ceiling. In the museum, you can see historic ceramic vases that once held herbs and products, as well as historic books and equipment. There is a tea room where visitors can taste teas and a museum store.
Today, production is highly mechanized. Outside Florence, near the Medici Villa della Petraia, the Officina Profumo-Farmaceutica di Santa Maria Novella has recreated a traditional hortus conclusus (enclosed garden) in the 13th-century tradition, using fresh herbs and ingredients to make its products.
Vera Koester
- Officina Profumo-Farmaceutica di Santa Maria Novella, Via della Scala 16, Florence, Italy
On to Austria to pay respects to a pioneer of thermodynamics and statistical mechanics …

Photo of Boltzmann’s grave: Dr. Bernd Gross, wikimedia commons, CC BY-SA 3.0 DE
20 Disorder Defined and Other Insights
Vienna, Austria
See the destination
Ludwig Eduard Boltzmann
Ludwig Eduard Boltzmann (1844 – 1906) was an Austrian physicist and philosopher. He his best known for his work on the development of statistical mechanics, and the statistical explanation of the second law of thermodynamics.
In 1877, Boltzmann provided the definition of entropy, or Boltzmann’s equation: S = k log W, where S is the entropy of an ideal gas, k is the Boltzmann constant, and W is the so called multiplicity (the number of real microstates corresponding to the gas’s macrostate). Simplified, the Boltzmann formula shows the relationship between the entropy and the number of ways the atoms or molecules of a system can be arranged.
Vienna Central Cemetery
When you are in Vienna, Austria, you can visit Ludwig Boltzmann’s grave on the “Zentralfriedhof” (Vienna Central Cemetery; Group 14 C, No. 1). The Vienna Central Cemetery was opened in 1874 and is one of the largest cemeteries in Europe, with an area of almost 2.5 km2 and around 330,000 gravesites with around three million deceased. It is located on the southeastern edge of town. The majority of the main cemetery has always consisted of Catholic graves. In addition, there are sections and cemeteries of other denominations: Buddhist, Protestant, Islamic (old, new and Islamic-Egyptian part), Jewish (old and new cemetery), Orthodox (Russian, Greek, Romanian, etc.), Mormon.
The grave of Ludwig Boltzmann shows a bust and Boltzmann’s entropy formula (pictured). Boltzmann is far from the only celebrity laid to rest in this cemetery. There are many historical graves and graves of honor, e.g., for Ludwig van Beethoven (Group 32 A, No. 29), Franz Schubert (Group 32 A, No. 28), and Johannes Brahms (Group 32 A, No. 26), but also other scientists such as Josef Loschmidt (Group 56 B, Row 2, No. 23).
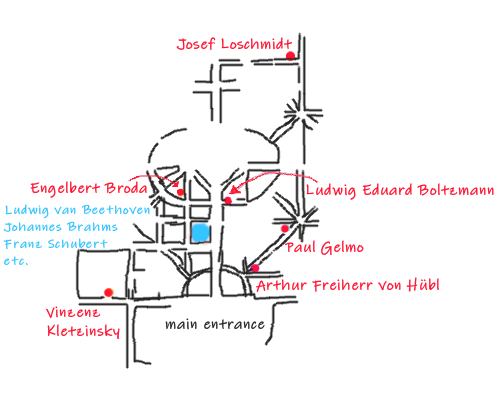 Other Chemists
Other Chemists
- Vinzenz Kletzinsky (1826 – 1882)
He was a brilliant speaker whose popular scientific lectures were very popular. As a chemist, he was primarily active in analytical work, and as such enjoyed a great, if sometimes controversial, reputation among physicians and technicians.
Group 29, row 1, no. 35 - Arthur Freiherr von Hübl (1853 – 1932)
He was a field marshal lieutenant, chemist, and cartographer, who played a decisive role in the modernization of cartography.
Group 12 D, Row 1, No. 11 - Paul Josef Jakob Gelmo (1879 – 1961)
He worked on synthetic dyes and discovered sulfanilamide (an organic compound consisting of an aniline derivatized with a sulfonamide group) in 1908. However, their antibiotic properties were discovered only in 1932.
Group 15 A, Row 1, No. 20 - Engelbert Broda (1910 – 1983)
He first worked at the Medical Research Council at University College London, UK, on the conversion of light into chemical energy, then at the Cavendish Laboratory, on radioactivity and nuclear fission. During this time, he intensively studied the work of Ludwig Boltzmann.
In 1947, he returned to the University of Vienna. His main scientific work “Evolution of the Bioenergetic Processes” was published in 1975. Broda became a member of the Pugwash movement, in support of nuclear disarmament, and worked to propagate the use of solar energy. Engelbert Broda was accused of espionage (code name “Eric”) and might have been the Soviet Union’s main source of information on American and British atomic bomb research.
Group 33 G, No. 70
Catharina Goedecke and Vera Koester
- Wiener Zentralfriedhof, Simmeringer Hauptstraße 234, Vienna, Austria
- Website of the Vienna Central Cemetery (in German)
It offers an audio guide to the cemetery that can be downloaded via a smartphone app. - Alphabetical list of honorary graves
If you know who you are looking for, it will help you to find the grave.
Continuing in the footsteps of a pioneer of very early genetics …

Photo of Abbey: Misa.jar, wikimedia commons, CC BY-SA 3.0
21 Mendel Museum
Brno, Czech Republic
See the destination
Mendel
Who does not know Gregor Johann Mendel (1822 – 1884), the Augustinian monk who discovered the basic principles of heredity and, in this context, is best known for his studies of plants, especially peas? He conducted his crossing experiments privately in the monastery garden over many years, following what was then a completely new approach with perseverance and an analytical approach, and founded the theory of heredity.
Mendel’s results were not understood in specialist circles for a long time. It was not until 1900, long after his death, that they were confirmed by other scientists who had obtained consistent results. Mendel also carried out other research, for example, in the field of beekeeping or apiculture.
The Museum
The Mendel Museum has been part of Masaryk University, Brno, since 2007 and aims to preserve the legacy of Gregor Johann Mendel. The museum on the grounds of the Augustinian Abbey in Old Brno enables scientists to meet the general public.
Visitors to the museum can see permanent and temporary exhibitions. One can see the monastery garden from the middle of the 19th century, where Mendel conducted his experiments. One can visit Mendel’s room, where he lived and worked until his appointment as abbot in 1868, and explore the monastery and its history. In the Baroque library, which contains some 27,000 volumes, visitors learn how faith and science can coexist.
The exhibition features original items that belonged to the “father of genetics”, such as his school reports, books, or teaching aids, and the book “Experiments on Plant Hybrids”. In addition to these antique exhibits related to Mendel’s studies, his monastic life, and scientific research, the exhibition uses audiovisual technology on glass panels to provide visitors with an interactive experience of Mendel’s work and life.
In addition, the Mendel Lecture Series offers lectures with outstanding personalities in the fields of biology, genetics, and molecular biology for a specialized audience and scientific events are organized in the Augustinian Abbey.
Vera Koester
- Mendel Museum, Mendlovo nám. 1a, 603 00 Brno-střed, Czechia
Another visit to the most famous female Nobel Laureate

22 Visiting Marie Curie
Warsaw, Poland
See the destination
This small, lovingly managed museum is located in the historical center of Warsaw. In 1967, on the occasion of the 100th anniversary of Maria Skłodowska-Curie’s birth, the Polish Chemical Society (Polskie Towarzystwo Chemiczne; PTC) founded the museum in the house where the Nobel Prize winner was born. Today, the PTC runs the museum jointly with the City of Warsaw.
The house has a huge entrance hall, but still you feel more like you are visiting than in a museum. Marie Curie’s mother, a teacher, ran a girls’ boarding house here.
The permanent exhibition consists of an extensive collection of photographs, letters, documents, personal items, and original furniture of the scientist and her family. Part of the exhibition is set up as the Curies’ Paris laboratory. The exhibits also include measuring instruments from the laboratory of Maria and Pierre Curie, as well as sculptures by famous Polish artists, stamps, medals, an d banknotes commemorating Marie Curie.
d banknotes commemorating Marie Curie.
On the picture above you can see one of the exhibits, a model of the Paris laboratory with Marie and Pierre Curie dolls.
The upper part of the house houses the offices of the Polish Chemical Society.
Vera Koester
- Maria Skłodowska-Curie Museum (Polish: Muzeum Marii Skłodowskiej-Curie; MMSC), 16 Freta Street (ulica Freta 16), Warsaw, Poland
Also of Interest
[1] Vera Koester, 100 Years Polish Chemical Society, ChemistryViews 2019. https://doi.org/10.1002/chemv.201900087
Time to try something yourself

23 Playing with Light, Electricity, Sound, and Planets
Tallin, Estonia
See the destination
There are many science museums, such as the Experimentarium (Copenhagen, Denmark), the Deutsches Museum (Munich, Germany; see above), VilVite (Bergen, Norway), and Heureka (Helsinki, Finland) – they are all great and fun to visit. In Estonia, there is the Energy Discovery Center, an exhibition for families and children with more than 130 hands-on exhibits on about 3,000 m² in a historic power plant.
It interactively presents topics such as electricity, lightning, renewable energy sources, nuclear energy, sound and optics, and classical physics. There are turbines and generators on display, which were used in the power plant. A highlight is the lightning demonstration with a Tesla coil in a Faraday cage. Lightning up to three meters high can be generated here, which they say is unique in Europe.
In a separate building there is a physics laboratory and a seminar room, to which there is an elevator which is about 100 years old. In addition, the museum offers scientific theater programs and workshops and has a planetarium.
Vera Koester
- Energy Discovery Center, Põhja puiestee 29, Tallinn, Estonia
We end our trip in Greece:
 Photo: © Spiros Kitsineli
Photo: © Spiros Kitsineli
24 Salt Production and Largest Salt Marsh in Greece
Messolonghi, Greece
See the destination
Salt Museum
If you want to know more about salt and its production and, at the same time, experience a unique landscape, visit the lagoon of Messolonghi. Here, about 65 % of the country’s salt is produced in two so-called salt pans that have been active for hundreds of years. The Salt Museum is located in the middle of one of these salt pans in a former dormitory of salt workers.
The museum shows where salt comes from and how different salts from all over the world are, for example, in color and grain. It also shows how salt is used (allegedly there are 14,000 uses), and how salt influenced health, but also literature, art, and religion. The visitor can see old tools used by workers in the salt pans, and the observatory offers a view of the entire saline.
Salt Production
Table salt is 97 to 99 % sodium chloride. Commercially, salt is sold with impurities, mainly potassium iodide, that is also essential in the diet. The sea contains salt at a rate of 2.7 to 3.8 %. In regions with a warm climate, this percentage increases, while it decreases in seas where many rivers flow. The Mediterranean Sea is one of the most saline seas.
The production season starts in March–April. The seawater is channeled into the preheaters of the salt pans, i.e., areas suitably configured for the storage and isolation of seawater. The water is heated and slowly dries up, while in specific conditions of density it is transferred from one heater to another. In this way, insoluble materials and unnecessary salts, which are produced during the drying of the sea, are removed. At the end of the summer, the salt-saturated seawater is transferred to the alopecias, or pans for the locals, where it is left to dry.
After collection, the salt is washed with brine to preserve its physical and chemical characteristics. What will come out is natural salt that retains its taste and about 88 trace elements, essential for health.
Messolonghi and What Else to See
Messolonghi is the capital of western Central Greece. The city, located 247 km west of Athens, is built on the alluvial arm formed between the lagoon of the same name and the lagoon of Kleisova.
The lagoons are also famous for their rare species of birds and fish, and you will find traditional fishermen’s huts built on wooden piles above the water. A trip to the small island of Troulida is very popular when the setting sun is reflected in the waters of the lagoon.
Spiros Kitsinelis
a chemist living in Athens, born in Messolonghi
- Salt Museum, Tourlida, Messolonghi, Greece
We hope you enjoyed our chemistry Europe tour as much as we did. Maybe we will travel to another continent together soon.
Maybe you have already visited one or more of these places or other destinations you would like to recommend. Please let us know in the comments below.
Also of Interest

Places around the world that we think are exciting for chemists to visit








I was a bit embarrassed by the DEC.02 ENTRY, SPAIN. The most interesting place over there is
showing a one-dimensional array of elements according to the hourly varying speculations of money-increase oriented people?
Could the author find no other place of chemists’s interest in whole Spain?
What about Cataluña’s University of Barcelona with ‘Aula García-Banús’ (with bifurcating Periodic Table after Antropoff) probably the last visible vestige of the residence of the Section of Chemistry at the historical University building that was left in 1969. Although I am no expert in the field of expertise of the respected author, I guess that there are many more places of historical interest for chemists in Spain. Maybe the author is more interested in the interests of Humanity-Arts-Social-Science (HASS) oriented people?
Thank you for suggesting Antonio García Banús’ Periodic Table from 1934 in classroom 111 of the Faculty of Philology at the University of Barcelona.
The 24 must-see places are, of course, a selection and are in no way meant to exclude that there are many more exciting places. We couldn’t cover every country and certainly don’t know everything, but we would also be happy if readers recommend additional places to these 24; if you like with a link, why you recommend visiting it, and how to visit it.
The old City Cemetery in Göttingen, Germany, is a historic graveyard that holds the graves of renowned scholars. Nine Nobel laureates are buried there:
Max Born (1882-1970), Physics 1954
Manfred Eigen (1927-2019), Chemistry 1967
Otto Hahn (1879-1968), Chemistry 1944
Max von Laue (1879-1960), Physics 1914
Walther Nernst (1864-1941), Chemistry 1920
Max Planck (1858-1947), Physics 1918
Otto Wallach (1847-1931), Chemistry 1910
Adolf Windaus (1876-1959), Chemistry 1928
Richard Zsigmondy (1865-1929), Chemistry 1925
In addition, well-known chemists such as Friedrich Wöhler (1800-1882) have also found their resting place.
Max Planck, for example, passed away at the age of 89 on October 4, 1947. At the base of his otherwise simple gravestone, the formula for Planck’s quantum of action is engraved.
Thank you for the additional recommendations of exciting places for chemists in Europe. Feel free to post more or send us ideas via email.
Museum of Chemistry in Várpalota, Hungary
The Museum of Chemistry is located in Thury Castle, a 14th-century building in Várpalota, Veszprém County, Hungary. Since 1963, this stunning centuries-old castle has served as the home of the museum. It showcases the history of Hungarian chemistry and the chemical industry, highlighting remarkable achievements and prominent figures in the field.
The museum’s permanent exhibitions are divided into four sections, tracing the history of chemistry from alchemy and early chemical technologies to the various periods of Hungarian chemistry and the chemical industry, to the role of measurement and computer science in the chemical industry, featuring displays of mechanical and electronic computers, an NMO spectrometer, a gas chromatograph, and a GIER computer—a significant milestone in computer applications.
Furthermore, the Museum of Chemistry houses the Collection of the History of Mining, dedicated to commemorating the coal mining industry in Várpalota.
In 1990, Thury Castle in Várpalota was recognized as a part of the National Heritage by the Committee of National Heritage.
Enrique Moles Historical Chemistry-Physics Laboratory – a typical laboratory of the 1930s
Instituto de Química Física Rocasolano, 119 Serrano Street, Madrid, Spain
The National Institute of Physics and Chemistry was established by the Board for the Expansion of Studies and Scientific Research (JAE) during the Silver Age of Spanish science in the early 20th century. It was a center of excellence for research in these fields and housed renowned researchers such as Blas Cabrera, Enrique Moles, Julio Palacios, and others. Initially funded by the Rockefeller Foundation, the institute had state-of-the-art laboratories, a well-equipped library, and advanced technical infrastructure such as the production of liquid air, generation and distribution of direct and alternating currents at low and high voltages, special laboratories isolated from vibrations and thermal oscillations. Although the institute was dissolved in 1939, the building now houses the Rocasolano Institute of Physical Chemistry (IQFR) of the Consejo Superior de Investigaciones Científicas (CSIC, Spanish National Research Council).
To commemorate its 90th anniversary, the historical laboratory “Enrique Moles” of Chemistry and Physics was inaugurated at the center. It recreates a typical laboratory from the 1930s and features partially restored original furniture and various setups, including glass components used by Enrique Moles for atomic weight determination and instruments for electrochemical experiments. The laboratory also includes an exhibition space showcasing scientific instruments.
Integral Form of Maxwell’s Equations on the Warsaw University Library
The integral form of Maxwell’s equations is etched in concrete on the side of the Warsaw University Library located at Dobra 56/66, 00-312 Warszawa, Poland.
We just learned on Mastodon that there is much more to see in Heidelberg: A stroll through the history of chemistry along the Hauptstrasse
Thank you for the great addition!