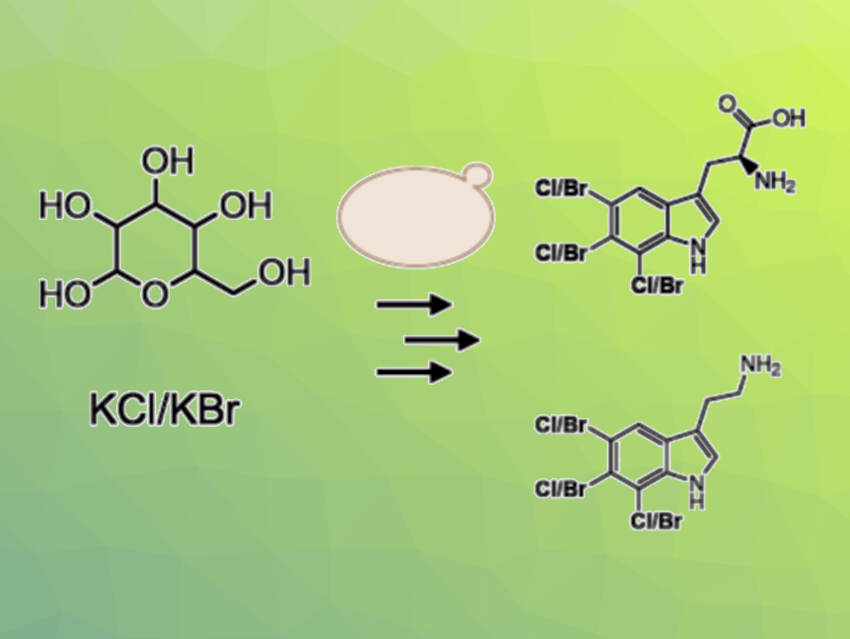
Indole scaffolds are often found, e.g., in bioactive heterocycles and natural products. Substituted indoles can have pharmaceutical or agrochemical applications. Regioselective halogenations of indoles can be useful to tune the properties of these compounds for different applications. However, chemical halogenation approaches can be expensive, have poor atom economy, lack regioselectivity, and/or generate hazardous waste. Biosynthesis could be a useful alternative.
Nicholas Milne, Technical University of Denmark, Lyngby, Denmark, and Octarine Bio ApS, Copenhagen, Denmark, Irina Borodina, Technical University of Denmark, and colleagues have engineered Saccharomyces cerevisiae (brewer’s yeast or baker’s yeast) to be used for the production of halogenated tryptophan and tryptamine derivatives (pictured above on the right). The team prepared recombinant yeast strains that can express enzymes for the targeted reaction, i.e., tryptophan halogenases together with a flavin reductase and a tryptophan decarboxylase.
These yeast strains can then be used for the production of halogenated tryptophan and tryptamine with chlorine or bromine. The team found that some tryptophan halogenases show substrate promiscuity and can also halogenate tryptamine directly and that the co-expression of two tryptophan halogenases can result in the production of dihalotryptophan. Overall, the work demonstrates the potential of Saccharomyces cerevisiae for the production of halogenated natural products.
- Engineering Saccharomyces cerevisiae for the de novo Production of Halogenated Tryptophan and Tryptamine Derivatives ,
Nicholas Milne, Javier Sáez‐Sáez, Annette Munch Nielsen, Jane Dannow Dyekjær, Daniela Rago, Mette Kristensen, Tune Wulff, Irina Borodina,
ChemistryOpen 2023.
https://doi.org/10.1002/open.202200266
Also of Interest

Yeast has been of vital importance in winemaking, baking, and brewing since ancient times