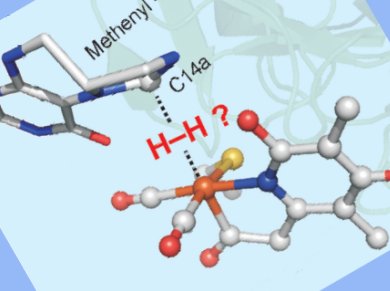
Microorganisms have been using H2 as a primary fuel source for billions of years. H2 uptake and evolution is regulated by the hydrogenase enzymes. Hydrogenases demonstrate highly efficient catalysis of the one-electron reduction of a proton. They differ in the shape, position and function of their active site, yet each carries an iron-carbonyl motif at their core.
The use of base metals by hydrogenases is an example of nature performing reactions under conditions that are more favorable than man-made equivalents — the current metal of choice for H2 fuel cells is the expensive and resource-limited platinum. As the need to find new ways to produce H2 from renewable source increases, hydrogenases offer a starting point for the design of catalysts for H2 production.
Topics surrounding all three classes of hydrogenases, the [FeFe]-, [NiFe]- and [Fe]-hydrogenases, are featured in a Cluster Issue of the European Journal of Inorganic Chemistry. The issue was edited by the guest editors, Marcetta Y. Darensbourg, Texas A&M University, USA, and Wolfgang Weigand, Friedrich-Schiller-Universität Jena, Germany.