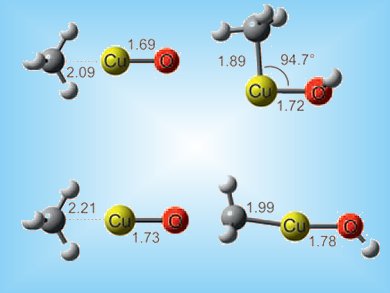
The activation of methane and its subsequent conversion into valuable feedstocks at ambient conditions could reduce its threat as a greenhouse gas and provide a route to bulk and fine chemicals. Bare [CuO]+ was predicted a decade ago to be a suitable, if not extremely powerful, candidate to convert methane to methanol, but it has never been sucessfully generated and studied experimentally.
Helmut Schwarz and colleagues, Technische Universität Berlin, Germany, have successfully generated the [CuO]+ cation in the gas phase. [CuO]+ was generated by laser desorption/ionization from isotopically pure copper 63Cu targets. It was shown to activate methane at room temperature both by hydrogen abstraction and by oxygen atom transfer. Mass spectrometry and DFT calculations revealed homolytic bond activation can be brought about by metal oxides with even numbers of electrons. These finding could lead to a new range of catalysts for the activation of methane under ambient conditions.
- Diatomic [CuO]+ and Its Role in the Spin-Selective Hydrogen- and Oxygen-Atom Transfers in the Thermal Activation of Methane
N. Dietl, C. van der Linde, M. Schlangen, M. K. Beyer, H. Schwarz,
Angew. Chem. Int. Ed. 2011.
DOI: 10.1002/anie.201100606