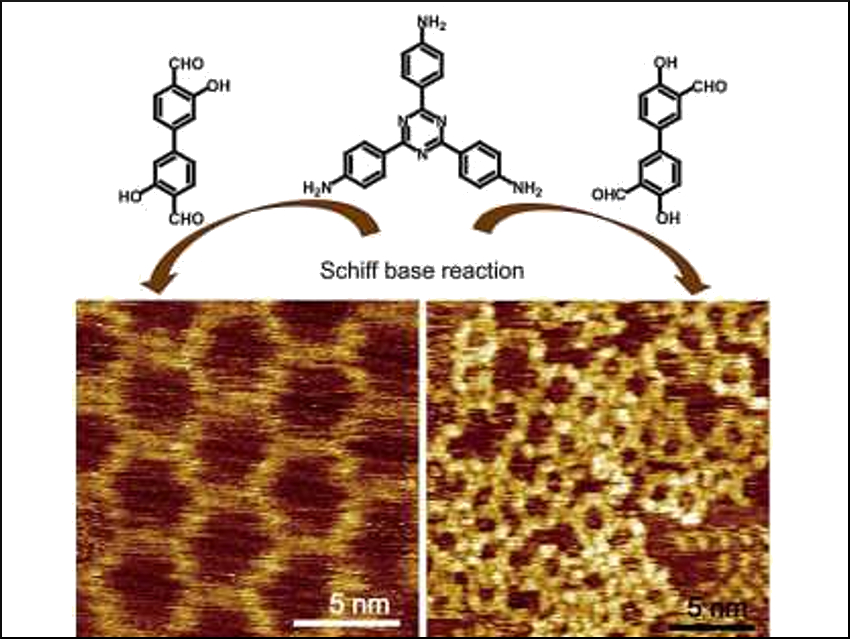
Two-dimensional polymers (2DPs) are materials with adjustable structures and tunable functional groups. On-surface synthesis—using a solid surface as the template—is an efficient way to construct 2DPs. However, preparing polymers with hydroxyl functional groups can be challenging with on‐surface synthesis due to side reactions.
Shengbin Lei, Harbin Institute of Technology and Tianjin University, both China, and colleagues have synthesized 2DPs and macrocycles functionalized with hydroxyl groups using surface-confined Schiff-base reactions on a graphite surface. The team used hydroxyl-functionalized aromatic aldehydes and aromatic amines as monomers to form imine polymers. A solution containing these precursors with a concentration of about 10−3 mol/L was deposited onto a freshly cleaved graphite surface by drop‐casting. The team then visualized the reaction with scanning tunneling microscopy (STM).
The researchers found that the reaction outcome can be steered from extended 2DPs to discrete macrocycles by changing the positions of the hydroxyl and aldehyde groups. The obtained 2DPs are molecular-scale films with well-ordered hydroxyl sites for chelating metal ions and may have applications in nanosensing.
- Surface Confined Synthesis of Hydroxy Functionalized Two-Dimensional Polymer: The Effect of the Position of Hydroxy Groups,
Lili Cao, Yanxia Yu, Xin Zhou, Shengbin Lei,
ChemPhysChem 2019.
https://doi.org/10.1002/cphc.201900360