William Francis Giauque was born on May 12, 1895, in Niagara Falls, ON, Canada. He studied chemistry at the University of California (UC), Berkeley, USA, where he received his Ph.D. in physical chemistry in 1922. He spent his entire career at UC Berkeley: as Associate Professor of Chemistry from 1922 to 1934, as Professor of Chemistry from 1934 to 1962, and as Emeritus Professor of Chemistry from 1962 until his death. Giauque died on March 28, 1982, in Berkeley.
Giauque’s most prominent work concerns entropy and the third law of thermodynamics. This law states that the entropy of a closed system approaches a constant value as its temperature approaches absolute zero. For an idealized, perfect crystal, the entropy at absolute zero (S0) is exactly zero: S0=0. The law was formulated by Walther Nernst in 1906 and is also known as Nernst’s theorem.
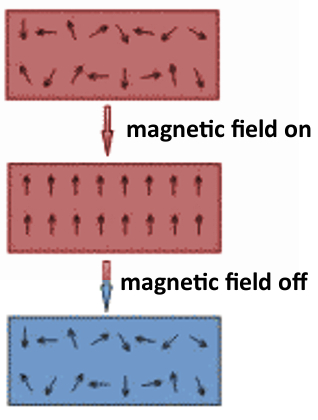 In 1927, Giaque developed a method in which a magnetic field is used to reach extremely low temperatures (well below 1 K). It can be used to provide evidence for the validity of the third law of thermodynamics. The method is called “adiabatic demagnetization” and was proposed independently by Peter Debye and William Giauque.
In 1927, Giaque developed a method in which a magnetic field is used to reach extremely low temperatures (well below 1 K). It can be used to provide evidence for the validity of the third law of thermodynamics. The method is called “adiabatic demagnetization” and was proposed independently by Peter Debye and William Giauque.
Adiabatic demagnetization involves applying a magnetic field while cooling a paramagnetic substance, e.g., in liquid helium. The substance’s magnetic dipoles are aligned in the magnetic field and their disorder is reduced (pictured right). Then, the liquid helium bath is removed to ensure an adiabatic, insulated process, and finally, the magnetic field is turned off. Energy cannot flow back into the system and thermal energy is transformed into magnetic entropy when the magnetic dipoles become disordered again.
With this method, the material can be cooled to temperatures as low as a few thousandths of one Kelvin. Based on this work, the Nobel Prize in Chemistry 1949 was awarded to Giauque “for his contributions in the field of chemical thermodynamics, particularly concerning the behaviour of substances at extremely low temperatures”.
Giauque’s research group performed many entropy determinations at low temperatures, e.g., on condensed gases. His work on the entropy of oxygen led to the discovery of the oxygen isotopes 17O and 18O in the Earth’s atmosphere. Previously, it had been thought that only one isotope, namely 16O, existed, and 16O has been used to define atomic masses. This discovery caused problems with the definition of the unified atomic mass unit: Chemists used the weighted average of the masses of known oxygen isotopes, while physicists used the mass of pure 16O. The definition of the unified atomic mass unit was changed to one based on the mass of 12C in 1961, which was then used by both chemists and physicists.
William Giauque is the answer to Guess the Chemist (101).
Sources
- William F. Giauque—Nobel Prize for Low-Temperature Research,
Marc A. Shampo, Robert A. Kyle, David P. Steensma,
Mayo Clin. Proc. 2006, 81, 587.
https://doi.org/10.4065/81.5.587 - Willian Francis Giauque (1895—1982): A Biographical Memoir,
Kenneth S. Pitzer, David A. Shirley,
National Academies Press, Washington D.C.,1996. - William F. Giauque: Biographical,
in Nobel Lectures, Chemistry 1942–1962,
Elsevier Publishing Company, Amsterdam, 1964.
Selected Publications
- Low temperature, chemical, and magneto thermodynamics: The scientific papers of William F. Giauque,
William F. Giauque,
Dover Publications, 1969.
ISBN: 978-096-473590-3 - Temperatures Below 1 Absolute,
William F. Giauque,
Charles Frederick Chandler Foundation, Columbia University, New York, 1936.
Also of Interest
- Overhaul of SI Units,
Vera Koester,
ChemViews Mag. 2018.
The mole and three other metric units redefined and linked to the values of physical constants - 150th Birthday: Walther Nernst,
ChemViews Mag. 2014.
Walther Nernst is well known for his work in the areas of electrochemistry and thermodynamics




