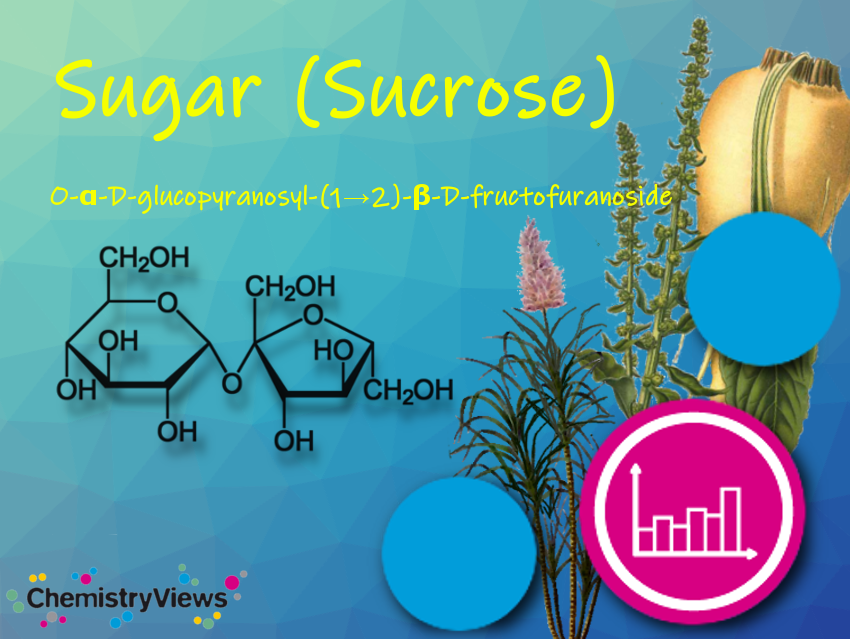
We all love sugar, but what is it chemically and since when do we know sugar? For our ancestors, the hunter-gatherers, sweet meant many calories. It was not until about 1850 that sugar became affordable and increasingly widespread.
Until the use of high-fructose corn syrup and artificial sweeteners, there was no real alternative to sugar. Honey was too expensive due to the complex extraction process.
.png)
.png)
Brown sugar gets its color from molasses. It is either an unrefined or partially refined sugar consisting of sugar crystals with a certain residual molasses content (natural brown sugar), or it is produced by adding molasses to completely refined white sugar (commercial brown sugar). In the latter case, it is easier to control the ratio of molasses to sugar crystals, and the production is cheaper. Sugar cane molasses is most commonly used because the flavor is generally preferred to beet sugar molasses.
Corn syrup is a cheap source of sugar. It is industrially produced by converting corn starch into sugars such as maltose, fructose, and glucose.
.png)
References
- Klaus Roth, Erich Lück The Saccharin Saga, ChemistryViews 2015. https://doi.org/10.1002/chemv.201500076
- William Cook, The Physiomedical Dispensatory, 1869. (Retrieved November 24, 2020)
Also of Interest
- Chemistry Advent Calendar 2020
ChemistryViews 2020.
Daily highlights from the chemistry of spices