The melting point is not an atomic property, it is a purely physical property. It is determined by the relationships in the crystal lattice. Therefore, the maxima and minima do not coincide with the beginning or end of a period as is the case with the atomic radii and ionization energies.
The graph shows that there are no more solid substances in the universe at temperatures above 3600 °C — only smelts, gases, and plasmas.
.jpg)
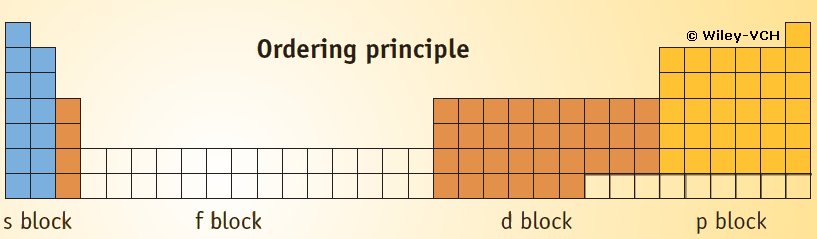
Taken from the book:
 World of the Elements, Elements of the World
World of the Elements, Elements of the World
Hans-Jürgen Quadbeck-Seeger
Wiley-VCH Verlag GmbH & Co, KGaA, Weinheim, 2007
Print ISBN: 9783527320653, Online ISBN: 9783527611577
DOI: 10.1002/9783527611577
Other Clever Picture dealing with Periodicity of Atomic Properties
- Ionic Radii of the Elements,
ChemViews Mag. 2011.
DOI: 10.1002/chemv.201000087
Ionic radii vary with coordination number, spin state, and other parameters but periodic trends can be recognized - Atomic Radii of the Elements,
ChemViews Mag. 2011.
DOI: 10.1002/chemv.201000080
Atomic radii of the main group elements vary in a predictable and explicable manner across the periodic table