Hydrogen Fuel
In the global hunt for sustainable fuels, hydrogen has much to offer. Not only is it the universally most abundant element, its energy-releasing combustion and electrolysis reactions with oxygen produce no greenhouse gases. Depending on the oxygen source, the only omission is water. In fuel cell technologies a simple electron exchange in the reaction of hydrogen and oxygen to water generates electricity. Applications of hydrogen fuel cells range from cars and airplanes, to portable fuel cell devices, emergency power systems and submarines that can “hold their breath” underwater for weeks. However, as a highly flammable gas with high potential for explosion in contact with air, its storage and transport can be impractical.
Hydrogen storage facilities that use compressed or liquid hydrogen require high pressure, thus, high energy input and myriad safety considerations. The adsorption of hydrogen onto supports, formation of metal hydrides, or incorporation of hydrogen into interim chemicals (chemical hydrogen storage) offer safe, diverse, and low-energy alternatives. After the chemical storage of hydrogen, it can be released as required.
Storing Hydrogen, Chemically and Safely
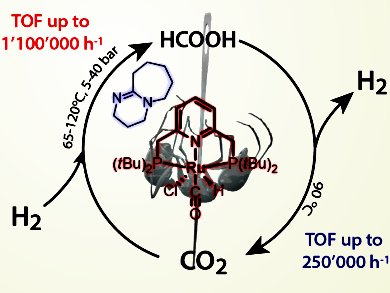
Chemical candidates for hydrogen storage include carbohydrates, ammonia, and ionic liquids. In particular, a hydrogen storage system using formic acid, first developed in 2006, has become a strong research interest. Carbon dioxide is hydrogenated by using pure hydrogen and a catalyst. A key benefit to the decomposition of formic acid for hydrogen release compared to other leading chemicals is the absence of carbon monoxide byproduct; only carbon dioxide and hydrogen are released. Catalytic formic acid hydrogenation systems also display high stability and longer catalytic lifetimes. For ultimate efficiency, both the hydrogen capture (carbon dioxide hydrogenation) and hydrogen release reactions are run with the same catalyst in one reaction vessel.
Enter Dr. Evgeny A. Pidko and co-workers from the Eindhoven University of Technology, The Netherlands: they have developed a catalytic system that can perform such storage/release cycles with unprecedented activity. The rates of both the storage and release mechanisms, as reported recently in ChemCatChem, are the highest known to date, furthermore, the system is particularly stable. This improves the prospects of a formate-based hydrogen battery. Integrated into a device, such batteries can replace high-pressure gas tanks by using hydrogen chemically bound in liquid, which is easy to store and transport.
Releasing the Potential
How to control storage and release? This system uses a simple temperature switch: hydrogen is stored at 65 °C under pressurized conditions and released after decompression and heating to 90 °C. This is enabled by the presence of a strong base that controls the mechanism of the reversible hydrogenation reaction. Other systems use, for example, a pH switch. The ruthenium metal center of the catalyst is bound to a PNP pincer ligand. Capture of hydrogen, its conversion to formate, followed by temperature-triggered formate dehydrogenation and hydrogen release, all occur at the metal center.
Some of previous systems that use an iridium catalyst employ similar ligands but with lower turnover numbers under much harsher conditions. Inspired by nature, one such iridium-based system relays protons between hydrogen bonds and added bases just like in the active site of certain enzymes.
Practical application of this catalytic system will require optimization of the hydrogen storage capacity. Other storage chemicals, for example methanol, may also show potential. Whilst noble metal catalysts are currently top in terms of performance in reversible carbon dioxide hydrogenation, iron and cobalt-based systems offer strong competition and provide a low-cost alternative.
Asking Dr. Pidko what further developments this research might lead to, he told ChemViews magazine that “in light of very recent literature on methanol reforming (to hydrogen and carbon dioxide), our paper may stimulate researchers to look at reaction mechanisms more carefully. The base promoter, often overlooked, turns out to be a useful tool for controlling the mechanism steering the hydrogen evolution reaction. We managed to improve the performance of our catalyst >50-fold and a great deal of it came from mechanistic analysis, done by theory and calculations rather than experiments. Many practical questions can be answered using theoretical knowledge like: Which solvent is better? Which gas proportions to use? How to speed up the reaction?”
Kyoko Nozaki, The University of Tokyo, Japan, has pioneered work in this field and describes why such advances are needed for hydrogen storage reactions: “For hydrogen storage, the hydrogenation/dehydrogenation process has been studied for several organic molecules, such as carbon dioxide/methanol and toluene/methylcyclohexane. In practical use, the reaction rate is essential and thus development of a catalyst system effective for both hydrogenation and dehydrogenation with the same base is highly desired.”
Continuing, she states that “the PNP ruthenium system developed by Pidko et al. shows high efficiency for both directions of the reversible process and thus paves a road towards a practical hydrogen storage technology”.
- Highly Efficient Reversible Hydrogenation of Carbon Dioxide to Formates Using a Ruthenium PNP-Pincer Catalyst,
Georgy A. Filonenko, Robbert van Putten, Erik N. Schulpen, Emiel J. M. Hensen, Evgeny A. Pidko,
ChemCatChem 2014, 6, 1526–1530.
DOI: 10.1002/cctc.201402119