Given the great number and wide range of biological activities they regulate, kinases are among the most important enzyme targets for drug development efforts, particularly in the field of oncology.
One of the main challenges faced by researchers in this area is inhibitor specificity; many compounds that effectively block the activity of a given target kinase also show significant inhibition of other kinases. Winssinger and colleagues have developed some promising fluoroenone lead compounds based on an entirely different molecular scaffold, the resorcylic acid lactones (RAL).
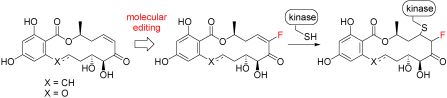
“The significance of this work does not lie solely in the chemistry and IC50 values, but also in the fact that there is a tremendous interest in irreversible kinase inhibitors that can be used to engineer kinase specificity and to profile kinase activity,” says Winssinger. “The cis-enone resorcylic acid lactones represent an alternative scaffold to the purines, which have been previously exploited. From a therapeutic perspective, these compounds are the first irreversible inhibitors of kinases involved in angiogenesis (VEGFRs and PDGFRs).”
- Molecular Editing of Kinase-Targeting Resorcylic Acid Lactones (RAL): Fluoroenone RAL
R. Jogireddy, S. Barluenga, N. Winssinger
ChemMedChem 2010, 5, 670.
 Cover Picture of ChemMedChem Issue 5/2010
Cover Picture of ChemMedChem Issue 5/2010
Nicolas Winssinger’s comment on why he chose the galley proof as a cover picture:
 “The phrase ‘molecular editing’ was coined by Professor Danishefsky in reference to the chemist’s ability to revise particular aspects of a natural product’s structure and hence properties. While natural products have been refined through cycles of selection/diversification, they are not always perfect pharmacological agents and editing their structure can be beneficial. Analogously, manuscripts go through a selection process but galleys can still benefit from editing.”
“The phrase ‘molecular editing’ was coined by Professor Danishefsky in reference to the chemist’s ability to revise particular aspects of a natural product’s structure and hence properties. While natural products have been refined through cycles of selection/diversification, they are not always perfect pharmacological agents and editing their structure can be beneficial. Analogously, manuscripts go through a selection process but galleys can still benefit from editing.”