The colors of the living world are extraordinarily diverse. In plants, most colors are due to pigments, whereas in animals they have either a pigmentary or a structural origin. The multitude of chromogenic mechanisms results from millions of years of evolution, during which pigments and structures have been selected for performing various tasks, some of them in relation to animal perception, and others without any link to this perception – or even to light itself.
Production, Diversity and Functions of Colors
The color of an object is the result of interactions between incident light and this object as well as between the light sent back by the latter and the visual system of an observer. The diversity of colors in the living world results essentially from differences in pigment composition and the surface microstructure of plants and animals. It is also dependent on the way the observer perceives colors, and this perception differs considerably depending on the particular animal. Finally, to a lesser extent, variations in the ambient light might play a role.
In this article, the origin of color diversity in the living world will be examined from the chemical and physical points of view. Then the discussion will examine the various functions of the colors of life as well as the ecological and evolutionary mechanisms that, by selecting these functions, induce the diversity of the pigment compositions and surface microstructures of plants and animals, and thus their colors.
I. The Production of Colors in Plants and Animals
Generally speaking, it is necessary to distinguish between the colors that depend on the chemical nature of an object and those that do not [1, 6]. In the first case, the selective absorption of light by pigments in certain ranges of wavelengths is the origin of the color perceived by our eyes, which collect the radiation that is not absorbed but instead reflected by the body. Sometimes, pigments are fluorescent: The fluorescence (emission of light following the absorption of light) then contributes to the colored perception, thereby making the colors brighter. In all these situations, we are talking about pigment colors that are present in plants as well as in animals.
In the second case, independently of the chemical nature, the physical phenomena such as refraction, scattering, interference, and diffraction of light are responsible for the observed colors. Here we are talking about physical colors as well as structural colors when the surface microstructure of the material is the origin. This case is common in animals but rare in plants.
Pigment Colors
Pigments in Plants
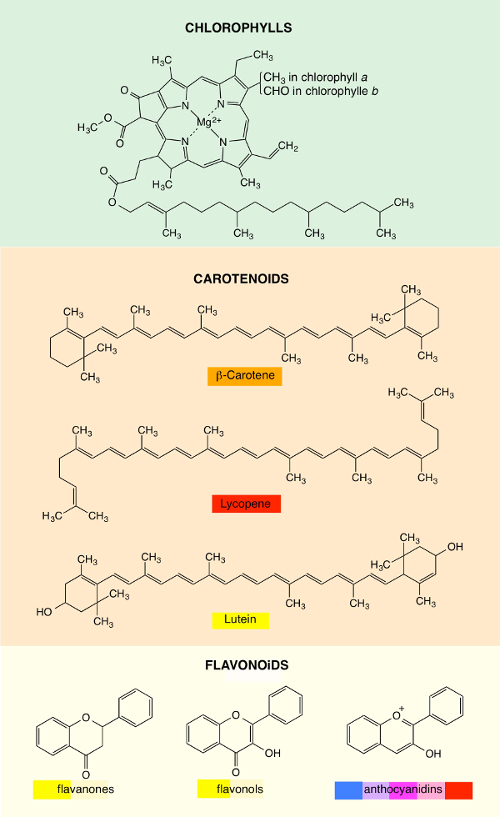
The colors of vegetables are due to various families of pigments that can exist simultaneously in the same plant. The most important three families are chlorophylls, carotenoids, and flavonoids [7–10].
Chlorophylls exist in diverse forms. The most widespread are chlorophylls a and b (Figure 1). They possess a porphyrin ring that contains a magnesium atom in the center, and a long chain with 20 carbon atoms is grafted onto this ring. They absorb sunlight at wavelengths shorter than 700 nm. However, the efficiency of absorption is weak around 530 nm, and in this wavelength region, light is sent back by diffuse reflection (that is, in all the directions), which provides to our eyes a general sensation of green. The various nuances of green result from the presence of several types of chlorophylls and other pigments, such as carotenoids and flavonoids, which contribute a little to the color.
Carotenoids, which are very widespread in the world of plants, provide yellow, orange, and red colors (Figure 1). In this family, we can distinguish between carotenes and xanthophylls. The first consist only of carbon and hydrogen atoms, whereas the second contain oxygen atoms in addition. The leader of carotenes is β-carotene, which is responsible for the orange color of carrots. As for tomatoes, they owe their red color to lycopene. The most important of the xanthophylls is lutein, which is yellow. It can be found in corn and especially in the green leaves of vegetables (e.g., spinach, sorrel, broccoli, cabbage), even if its color is masked by chlorophylls.
|
|
|
Figure 1. Examples of plant pigments that belong to the chlorophyll, carotenoid, and flavonoid families. For the latter, the hydroxyl groups (OH) in various positions are not shown. |
Flavonoids are especially present in flowers. They are flavonic compounds that belong to the polyphenol class. The skeleton consists of three cycles (two benzene rings and an oxygenated heterocycle) that bear hydroxyl groups (OH) in various positions. They can be divided into several groups of compounds, in particular, flavanones, flavonols, and anthocyanidins (Figure 1). Flavanones and flavonols give various shades of yellow. Anthocyanidins undergo a glycosylation by an enzyme within plants, which leads to anthocyanins. Their colors strongly depend on the number and on the position of the hydroxyl groups: red, pink, blue, violet, and purple.
These colors can be affected by complexation of metals or by changes in acidity. For example, the color of cyanidin is blue in basic medium (e.g., cornflower), violet in neutral medium, and red in acidic medium (e.g., poppy). Red cabbage also owes its color to cyanidin. Its juice is blue in basic medium and red in acidic medium. Anthocyanins are also responsible for the color of dark red fruits (e.g., blueberries, blackcurrant, black grapes).
Animal pigments
It is important to distinguish between pigments that animals produce by themselves (e.g., melanin, pterins, ommochromes, bile pigments) and those that are foodborne (carotenoids) [7]. Among the pigments produced by animals, the most common are melanins. They are usually brown to black, but also range from yellow to reddish brown. They are found in association (or not) with other pigments, particularly in the hair of mammals, bird feathers, butterfly wings, and so forth. If melanin production should fail, the animal is white with red eyes (albinos). When melanins do not migrate into the hair or feathers, the animal has a white coat or feathering, but pigmentation of the eyes, the skin, and the legs is normal. Finally, excessive melanin production gives a black color or something close to it (e.g., rabbits, dogs, hamsters, cats, panthers). Butterflies have very different colors on account of various pigments, in particular, pterin and ommochromes [11, 12] (Figure 2).
|
|
|
Figure 2. The colors of the peacock butterfly (Inachis io) are due to various pigments – except the blue color of the eyespots (right), which is of interference origin (© B. Valeur). |
Animals cannot produce carotenoids. Those they do possess have been eaten and often processed by the body. Birds are particularly capable of performing the processing of carotenoids. Many of them exhibit very diverse colors from yellow to red: goldfinch, robin, tit, chaffinch, and so forth. [13, 14]. As for the pink flamingo, its color exclusively arises from its diet, which consists mainly of planktonic crustaceans that feed on algae which are rich in carotenoids converted to red astaxanthin and canthaxanthin.
Note that contrary to plants, the presence of blue and green pigments is exceptional in animals. Some rare butterfly wings contain such pigments. In most cases, the blue is of a structural type, as we shall see below. As for the green, it most often results from a blue structural color superimposed over the yellow of a pigment. This is particularly true for the skin of frogs and the green feathers of birds.
Structural Colors
Iridescence
Many insects exhibit colors that change depending on the angle of observation. This phenomenon, called iridescence, is due to light interference arising from a periodic structure with a pitch that has an order of magnitude comparable to the wavelengths of light [3, 7, 11, 12].
For example, the carapace (the correct term is cuticle) of beetles consists of several superimposed thin layers. A light wave that reaches the surface is partially reflected on the various layers, and the light waves that emerge from the front side interfere with each other. According to the local thickness and the angle of observation, the intensity of light at certain wavelengths is reduced, whereas it is reinforced at other wavelengths. Hence, the emergence of colors.
The metallic blue of the Morpho butterfly is also of interference origin. However, unlike the previous case, it is not multilayered. Instead, it stems from striations that consist of a stack of 6 to 12 chitin lamellae maintained at a constant distance [11, 12]. These grooves (about 1800 per mm) form a diffraction grating that reflects light only for wavelengths below about 500 nm (i.e., more or less blue).
The interference colors due to the presence microlamellae also exist in the feathers of peacock, pheasant, and many other birds, from the exotic ones to the most common [13, 14].
Colors by Light Scattering
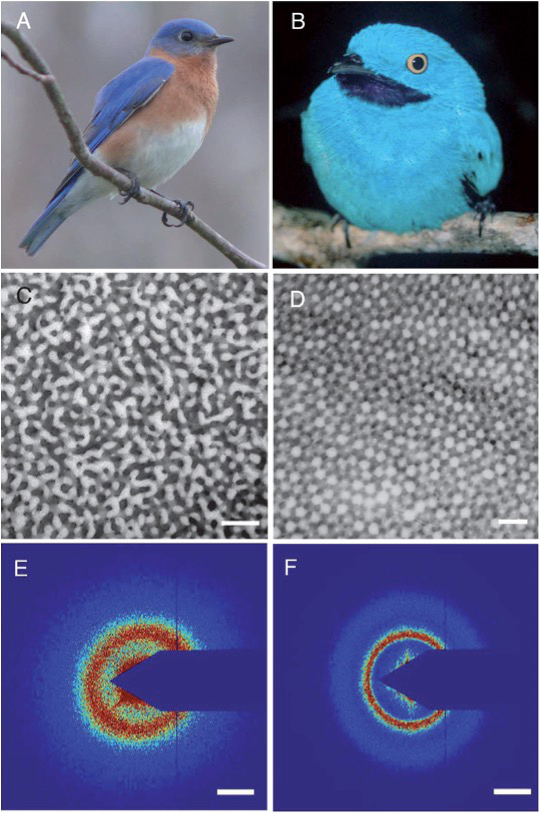
The blue color of feathers deserves special attention because it is not due to blue pigments and the colors do not change according to the observation angle, unlike the wings of Morpho butterflies. Therefore, there is no iridescence. Another physical phenomenon is actually involved: Light scattering by structural elements of very small size (several tens of nanometers) that constitute the feather barbs.
For a long time, it was thought that the color was due to Rayleigh scattering (i.e., light scattering by particles with sizes less than one-tenth of the wavelength of light), a phenomenon responsible in particular for the blue color of the sky. However, this explanation is insufficient for bird feathers, as electron microscopy reveals that the feather barbs are made of keratin and air nanostructures with some partial order (Figure 3) [15]. Thus, constructive interferences occur locally without iridescence (i.e., without color changes depending on the observation angle) because there is no order over long distances. The interference reinforces the intensity of the scattered light at certain wavelengths that depend on the size and spacing of the elements that make up the nanostructure. It is, therefore, clear in this case that it is coherent light scattering.
|
|
|
Figure 3. The blue feathers of these birds owe their color to light scattering by nanostructures that contain keratin and air [15]. Reproduced with permission from The Royal Society of Chemistry |
For the male Eastern Bluebird (Sialia sialis) (Figure 3, A), electron microscopy reveals channel-type β-keratin and air nanostructures from back contoured feather barbs (C). For the male Plum-throated Cotinga (Cotinga maynana) (B), electron microscopy reveals spherical β-keratin and air nanostructures from back contoured feather barbs (D). Figure 3, E shows small-angle X-ray scattering data from channel-type feather barb of S. sialis, and F data from a spherical feather barb of C. maynana. (Scale bars (C–D) 500 nm, (E–F) 0.025 nm–1 of spatial frequency.)
Note finally that structural colors also exist in plants but with more rarity [7, 10]. Some have interference colors owing to multilayers. This is the case of the leaves of the tropical fern Danaea nodosa as well as the fruits of the African herbaceous plant Pollia condensata, the blue color of which is one of the strongest observed in the living world. As for the blue pine or Colorado spruce (Picea pungens ‘Glauca’), it owes its blue color to light scattering by fine particles of wax covering the needles.
II. Functions and Diversity of Colors
The various colors produced by the previously described mechanisms might have no specific function and thus are the mere byproduct of the chemical composition and surface microstructure of an object. Many colors, however, have been selected through evolution with regard to specific functions. We can identify two types of functions depending on whether an animal perceiving colors is intended or not. We will use the term “chromatic functions” for the first type and “achromatic functions” for the second. Yet one should keep in mind that, strictly speaking, the term achromatic function is nonsensical because, in life science, a color is defined as a sensation and thus color necessarily implies perception. For the ease of understanding, we will nevertheless engage in this misuse of language.
Achromatic Functions
Chlorophylls are the primary example of pigments selected for their achromatic function. They are present in all photosynthetic organisms including plants, algae, and bacteria. They are associated with proteins and gathered in light-harvesting antennae whereby excitation energy is transferred to a reaction center. In the latter, a special pair of chlorophylls a induces a charge separation and triggers a series of electron-transfer reactions [9]. The net effect is the oxidation of water and the reduction of carbon dioxide, which leads, by following the Calvin cycle, to the production of carbohydrates.
The absorption of light by pigments can also be used for filtering out parts of the light spectrum that are harmful to living organisms, mainly ultraviolet (UV) radiation. For example, the red coloration of leaves in autumn is caused by anthocyanin pigments used as a sunscreen when nutrients are released from the leaves to the trunk and the roots. In animals—mammals in particular—this sunscreen effect is achieved by melanin pigments; their concentration in skin tissue increases with light intensity. Thermoregulation is another type of protection against light. Dark colors are typically known to efficiently convert light energy into heat. Yet the link between irradiance and the degree of melanism can be relatively complex, because it is further influenced by other co-factors such as the body size and the type of physiological mechanism used to keep the body temperature constant [16].
In plants, pigments such as carotenoids and flavonoids are also powerful antioxidants owing to their ability to neutralize the free radicals that accelerate cellular aging. In some species, individuals can adapt their feeding strategy to ingest carotenoid-rich foods during periods when the intracellular concentration of free radicals is the highest [17].
Another type of protection relies on the mechanical properties of pigments. In many species of birds, for example, the trailing edge of the wings and tail tip are black. The high concentration of melanin pigments in such black terminal bands increases the resistance of feathers to abrasion caused by airborne particles [13].
Lastly, many pigments confer a chemical protection against pathogens or predators. In birds, once again, melanin slows down feather degradation caused by bacteria [18]. Similarly, anthocyanins have been shown to protect fruits against fungi [19] and the vegetative parts of plants against herbivores [20].
Chromatic Functions
Perception and Color Conspicuousness
By definition, the chromatic functions of colors are tightly linked to the ability of animals to perceive colors. Color vision requires at least two types of photoreceptors that differ in their wavelength of maximal sensitivity. Animals with a single type of photoreceptor can only perceive variation in light intensity. By comparing the relative stimulation of at least two types of photoreceptors, it becomes possible to extract information on the spectral composition of light.
Whereas most mammals use two types of photoreceptors to perceive colors, Old World primates and insects use three, and birds use four. Some butterfly species have eight distinct types of photoreceptors. The highest number of photoreceptors is found in mantis shrimp (Crustacea, Stomatopoda), which have up to sixteen types of photoreceptors – including twelve used for color vision [21, 22].
One of the most important chromatic functions of colors is to increase the likelihood of being detected by other organisms. Whether they are designed to attract pollinators, seed dispersers, or potential sexual partners, conspicuous colors that maximize the chromatic contrast with the environment frequently increase the fitness of the signaler – that is, the probability that it will transmit genes to the next generation. For example, one experiment showed that the chromatic contrast between artificial flowers and the background was negatively correlated with the time needed for bees to visit these flowers [23]. A pollination system based on conspicuous signaling colors creates a win–win situation because pollinators can visit and collect food from many flowers if they detect them rapidly, and plants can increase their own fitness if they have pollen transferred to many others (Figure 4). In this context, emission of fluorescence, luminous colors, sometimes makes an additional contribution to attractiveness.
|
|
|
Figure 4. Flowers attract pollinating insects on account of their colors, but also on account of their shape and fragrance (© B. Valeur). |
How do the diversity of colors, color perceptions, and conspicuousness relate to each other? One could hypothesize that color diversity originates from the huge variation in selection pressures imposed by perceivers across space and time. Indeed, we have already seen that different types of photoreceptors occur throughout the animal kingdom, and thus that animal species differ in their perception of colors. Moreover, color perception is not only determined by photoreceptors but is also influenced by other sensory modalities and by past experiences stored in memory. Thus, a color is unique to a given observer at a given time. However, natural color signals have evolved primarily through the effect of selection over successive generations of individuals and are thus adapted not to the specific perception of one individual but to the average perception that is representative of an entire population or species.
In addition, it is traditionally recognized that the influence of sensory and perceptual mechanisms that occur beyond the receptor level on the diversity of color perception is minute compared to that of the diversity of photoreceptor equipment [22]. With the exception of a few animal groups, such as butterflies and fish, photoreceptor equipment is quite conservative phylogenetically: Virtually all bee species have the same set of photoreceptors, as do all reptiles, all primates, all birds, and so on [21]. Ultimately, the diversity of visual systems and the adaptation of color signals to increase detection by these systems cannot alone explain the huge diversity of natural colors.
Beyond Conspicuousness
Natural colors do not only facilitate detection, they can also enhance recognition. For instance, species of plants that co-occur in a meadow tend to differ in coloration more than should be expected by chance [24]. This could be explained by a mechanism called “ecological sorting,” whereby a species colonizing a new site needs to draw pollinators’ attention while also facilitating their recognition and memorization for reproducing. Indeed, if a pollinator mistakes a rare, newly arrived plant species with a more common one, the former species will receive pollen only from the latter species and will thus fail to reproduce.
Recognition of conspecifics also promotes diversification of species through the course of their evolution. Hybridization is a phenomenon that mixes the gene pools of two recently diverged species, potentially combining both species into a single one. By facilitating species recognition, distinctive color signals can reduce hybridization and thus facilitate speciation. This has been evidenced in African starlings, a group of birds with many different species characterized by iridescent, vividly colored plumage. Through the course of evolution, the appearance of melanosomes with innovative forms and structures has allowed the creation of novel colors that could not be produced with the ancestral melanosomes present in these birds [25].
Interestingly, bird lineages with these new melanosomes have radiated into more distinct species than lineages that retained their ancestral melanosomes. Furthermore, species with the new melanosomes have a tendency to be more social than other species, which illustrates how an innovation in a color-production mechanism can relax constraints on speciation owing to species recognition.
Selection by Multiple Perceivers
Most flowers and fruits are “generalists,” which means that they are visited by several species of pollinators and frugivorous animals. These color signals thus have to be adapted to attract multiple perceivers characterized by distinct color vision [26]. But even in specialized interactions, signals are usually perceived by third parties. The selection by unintended perceivers explains why sexual signals frequently evolve in private communication channels, which allow the attraction of females while minimizing detectability to predators.
In Müllerian mimicry, for example, several noxious species mimic each other to facilitate their categorization as “unpalatable organisms” by potential predators. This communication strategy still implies that, unlike predators, individuals are able to recognize conspecifics, at least for mating. In Heliconius butterflies, several species have evolved the ability to produce a new yellow pigment, 3-hydroxy-L-kynurenine (3-OHK), which reflects ultraviolet radiation [27]. Notably, the photoreceptor sensitive to UV was duplicated at the same time as the appearance of the 3-OHK pigment during the evolution of this species.
Having two photoreceptors sensitive to UV allows refined discrimination capacities in this spectral range, which has permitted the evolution of private, ultraviolet communication signals. Overall, it is well documented that the community of species surrounding a signaler generates a myriad of both mutualistic and antagonistic interactions that contribute to diverse communicative colors in plants and animals.
Heterogeneity of Visual Environments
Variation in ambient light modifies the perception of colors. On the evolutionary timescale, these variations can also drive the evolution of colored signals that are adapted to a specific environment. There is a great deal of evidence of such adaptations in water environments. While ambient light varies strongly with the depth of the water column, species of fish that live at a given depth tend to display coloration that allows them to be conspicuous amid the local ambient lighting [28].
Red fluorescence, for example, is quite common in the range of water depth that lacks red radiation. Fish with red patches thus appear highly conspicuous in these all-blue environments [29]. In land habitats, ambient light typically varies less than in water habitats (see, however, [30]); but here variations in the background coloration were also found to drive diversification of color signals [31].
The Multifunctionality of Colors
Any selection force that acts on the signaling function of a color-producing mechanism inevitably influences other functions linked to this mechanism. For example, flowers of the wild radish Raphanus sativus occur in two color morphs, violet or white, depending on the presence of anthocyanin pigments. Knowing that white flowers are greatly preferred by pollinators, which, therefore, visit and propagate more pollen of this color morph, how can we explain the perpetuation of violet flowers in natural populations of wild radish?
Experiments revealed that violet flowers are more resistant to attacks by herbivorous animals (e.g., caterpillars, slugs) than white flowers because violet ones accumulate more toxic secondary compounds (indole glucosinolate) [20]. In wild radish, therefore, the antagonism between selective forces that influence either reproduction or survival has allowed a long-term perpetuation of color polymorphism.
Emitting Colored Lights
All of the colors described so far result from the interaction between solar light and living organisms. At dusk, flowers, butterflies, and birds are seen as variations of gray shades before disappearing completely as light vanishes. Some organisms are nevertheless capable of producing colors even in darkness. This is due to bioluminescence [7, 32].
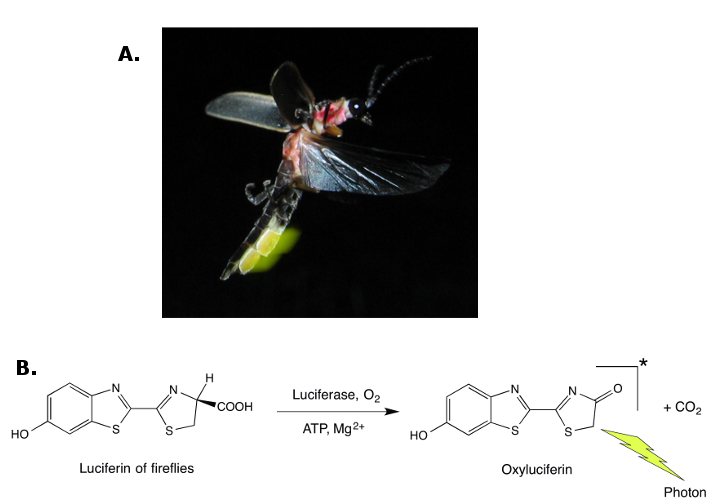
Bioluminescence is an emission of light that accompanies a biochemical reaction in which luciferin (a generic name) is oxidized by a specific enzyme called luciferase. The colors of emitted light are diverse; they range from blue to red and depend on the luciferin/luciferase pair. Bioluminescence has been observed in thousands of plant and animal species. It is quite uncommon among land organisms (mainly in beetles and fireflies), but it is relatively ubiquitous in the oceans (among crustaceans, jellyfishes, fishes, mollusks).
As with fluorescence, in most cases the function of bioluminescence remains unknown. There are a few species, however, in which the benefits of emitting light have been clearly identified: namely, increasing attraction of potential mates (Figure 5) or food items, and protection against predators [7, 32]. Occasionally, a bioluminescent organ can play several of these roles in the same animal.
|
|
|
Figure 5. Fireflies (here Photinus pyralis) are beetles with a bioluminescent abdomen (A). The yellow-green flashes of light they emit are communication signals between males and females for mating purposes (© Terry Priest, License: Creative Commons Attribution-ShareAlike). (B) The overall oxidation reaction of the luciferin of fireflies that leads to oxyluciferin is catalyzed by luciferase and requires adenosine triphosphate (ATP) and magnesium ions. |
Conclusion
The colors of the living world are produced by a wide variety of mechanisms. One of the main functions of the pigments is protection against sunlight and, in particular, the ultraviolet radiation that is harmful to living organisms. In the marine environment, where ultraviolet radiation is heavily filtered, there is a wide range of colors under white light that probably reflects a relaxation of selection forces on the protective function of the pigments. In many cases, the phenomena of fluorescence and bioluminescence make contributions and are all adaptations to a predominantly blue and dark environment. Life under ambient air has significantly constrained color diversity by selecting the pigments for their ability to collect light energy and initiate its conversion into chemical energy (through chlorophylls) or offer protection from sunlight. Thereafter, the colored signals certainly helped to diversify the natural colors on the surface of the earth.
The diversity of visual systems alone does not explain the diversity of colors and the mechanisms that produce them. The spatial and temporal variations under ambient light and the visual environment in general, the cognitive abilities of animals that enable them to store more and more stimuli, the environmental context and in particular the conflicts of interest between different observers of a color signal, the many links between the chromatic properties of color-production mechanisms and other physiological functions that are closely related to the selective value of an organism – these are the joint reasons for the evolution towards to the bright color palette of the living world that we admire today.
References
[1] B. Valeur, La couleur dans tous ses éclats (in French), Belin, 2011. ISBN: 978-2-7011-5876-1
[2] B. Valeur, La genèse des couleurs: un dialogue entre lumière et matière (in French), L’Act. Chim. 2015, 396, 29–33. Link
[3] M. Elias, J. Lafait (eds.), La Couleur. Lumière, vision et matériaux (in French), Belin, 2006. ISBN: 978-2-7011-4192-3
[4] K. Nassau, The Physics and Chemistry of Colour: The Fifteen Causes of Colour, John Wiley & Sons, New York, 2001. ISBN: 978-0-471-39106-7
[5] L. Zuppiroli, M.-N. Bussac, Traité des couleurs (in French), Presses polytechniques et universitaires romandes, Lausanne, 2011. ISBN: 978-2-88074-868-5
[6] R. M. Christie, Colour Chemistry, Royal Society of Chemistry, 2013. ISBN: 978-0-85404-573-0
[7] B. Valeur, E. Bardez, La lumière et la vie. Une subtile alchimie (in French), Belin, 2015. ISBN: 978-2-7011-7688-8
[8] J. Jeanfils, Pigments et biosphère. Les couleurs de la vie (in French), Vuibert, 2008. ISBN: 978-2-7117-4024-6
[9] P. Mathis, Pourquoi les feuilles sont-elles vertes? Les Petites Pommes du Savoir (in French), Le Pommier, 2003. ISBN: 978-2-7465-0125-6
[10] D. Lee, Nature’s palette. The Science of Plant Colour, Chicago Press, 2007. ISBN: 978-0-226-47053-5
[11] S. Berthier, Les couleurs des papillons ou l’impérative beauté. Propriétés optiques des ailes de papillons (in French), Springer, Paris, 2000. ISBN: 978-2-287-59657-5
[12] S. Berthier, Iridescences. The Physical Colors of Insects, Springer, 2007. ISBN: 978-0-387-34120-0
[13] G. E. Hill, Bird Colouration, National Geographic, 2010. ISBN: 978-1-4262-0571-2
[14] M. Pomarède, La couleur des oiseaux et ses mystères (in French), Armand Colin, 1990. ISBN: 978-2-200-37196-8
[15] E. R. Dufresne et.al., Soft Matter 2009, 5, 1792–1795. DOI: 10.1039/B902775K
[16] S. Clusella Trullas et al., J. Therm. Biol. 2007, 32, 235–245. DOI: 10.1016/j.jtherbio.2007.01.013
[17] D. Constantini, in Oxidative Stress and Hormesis in Evolutionary Ecology and Physiology, Springer, 2014, 111–142. DOI: 10.1007/978-3-642-54663-1_4
[18] G. Goldstein et al., The Auk 2004, 121, 656–659. DOI: 10.2307/4090304
[19] H. M. Schaefer et al., Natural Prod. Comm. 2008, 3, 1267–1272. Link
[20] R. E. Irwin et al., Ecology 2003, 84, 1733–1743. DOI: 10.1890/0012-9658(2003)084[1733:TROHIT]2.0.CO;2
[21] D. Osorio, M. Vorobyev, Vision Research 2008, 48, 2042–2051. DOI: 10.1016/j.visres.2008.06.018
[22] A. Kelber, D. Osorio, Proc. Roy. Soc. B: Biol. Sci. 2010, 277, 1617–1625. DOI: 10.1098/rspb.2009.2118
[23] J. Spaethe et al., Proc. Nat. Acad. Sci. USA 2001, 98, 3898–3903. DOI: 10.1073/pnas.071053098
[24] A. Gumbert et al., Proc. Roy. Soc. B: Biol. Sci. 1999, 266, 1711–1716. DOI: 10.1098/rspb.1999.0836
[25] R. Maia et al., Proc. Nat. Acad. Sci. USA 2013, 110, 10687–10692. DOI: 10.1073/pnas.1220784110
[26] J. P. Renoult et al., New Phytol. 2014, 201, 678–686. DOI: 10.1111/nph.12539
[27] S. M. Bybee et al., Am. Nat. 2012, 179, 38–51. DOI: 10.1086/663192
[28] M. E. Cummings, Evolution 2007, 61, 530–545. DOI: 10.1111/j.1558-5646.2007.00047.x
[29] M. G. Meadows et al., Proc. Roy. Soc. B: Biol. Sci. 2014, 281, 20141211. DOI: 10.1098/rspb.2014.1211
[30] D. Gomez, M. Théry, Ecol. Lett. 2004, 7, 279–284. DOI: 10.1111/j.1461-0248.2004.00584.x
[31] J. Ng et al., Evolution 2013, 67, 573–582. DOI: 10.1111/j.1558-5646.2012.01795.x
[32] T. Wilson, J. W. Hastings, Bioluminescence: Living Lights, Lights for Living, Harvard University Press, Cambridge, MA, USA, 2013. ISBN: 978-0-674-06716-5
Julien P. Renoult
Institut Arts, Créations, Théories et Esthétiques (UMR 8218), Université Paris 1 Panthéon-Sorbonne, 47 rue des Bergers, 75015 Paris, France. [email protected]
Bernard Valeur
Conservatoire national des arts et métiers, Département CASER, 292 rue Saint-Martin, 75003 Paris, France. [email protected]
The article has been published in French in:
- Les couleurs de la vie : mécanismes de production, fonctions et diversité, L’Actualité Chimique 2015, 397–398 juin-juillet, 12–18.

.jpg)