Pfizer announced very encouraging interim results of the Phase 2/3 EPIC-HR (Evaluation of Protease Inhibition for COVID-19 in High-Risk Patients) trial of its novel oral antiviral COVID-19 candidate PAXLOVID™ (PF-07321332; ritonavir).
At the recommendation of an independent Data Monitoring Committee and in consultation with the U.S. Food and Drug Administration (FDA), Pfizer will discontinue further enrollment into the study due to the overwhelming efficacy of these results. The company plans to submit the data as part of its ongoing rolling submission to the U.S. FDA for Emergency Use Authorization (EUA) as soon as possible.
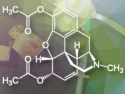
Treatment with PAXLOVID™
PAXLOVID™, developed at Pfizer’s laboratories, would be the first oral antiviral of its kind. It is intended to be prescribed as an at-home treatment at the first sign of infection or upon first awareness of exposure to avoid severe illness that can lead to hospitalization and death. According to the company, the drug has the potential as a therapeutic for several types of coronavirus infections.
PAXLOVID™ is an antiviral therapy with a SARS-CoV-2 protease inhibitor. It was designed to block the activity of SARS-CoV-2-3CL protease, an enzyme that the coronavirus needs to replicate. PF-07321332 inhibits viral replication at a stage known as proteolysis, which occurs prior to viral RNA replication. Co-administration with a low dose of ritonavir (an antiretroviral medication used along with other medications to treat HIV/AIDS) helps slow the metabolism or breakdown of PF-07321332 so that it remains at higher concentrations and active in the body for longer and helps fight the virus.
Phase 2/3 EPIC-HR Study
The randomized, double-blind study was conducted on non-hospitalized adult patients with COVID-19 who are at high risk of progressing to serious illness. Patients treated within three days of onset of symptoms (primary endpoint) showed an 89 % reduction in the risk of COVID-19-related hospitalization or death compared to patients who received a placebo. 0.8 % of patients who received PAXLOVID ™ were hospitalized by day 28 after randomization (3/389 hospitalizations with no deaths). By comparison, 7.0 % of patients who received a placebo were hospitalized or died (27/385 hospitalizations with seven subsequent deaths). The statistical significance of these results was high at p < 0.0001, the company said.
Similar reductions in COVID-19-related hospitalization or deaths were seen in patients treated within five days of onset of symptoms. 1.0 % of patients who received PAXLOVID ™ were hospitalized by day 28 after randomization (6/607 hospitalized with no deaths), and 6.7% of patients who received a placebo (41/612 hospitalized with ten subsequent deaths) (statistical significance p <0.0001). No deaths were reported in patients receiving PAXLOVID ™ in the entire study population up to day 28. In the placebo group, there were 10 (1.6%).
The Phase 2/3 EPIC-HR study began enrollment in July 2021 following positive Phase 1 clinical trial results. The Phase 2/3 EPIC-SR (Evaluation of Protease Inhibition for COVID-19 in Standard-Risk Patients) and EPIC-PEP (Evaluation of Protease Inhibition for COVID-19 in Post-Exposure Prophylaxis) studies began in August and September 2021, respectively. They were not included in this interim analysis and are ongoing.
- Pfizer Inc., New York, NY, USA
Also of Interest
- Collection: SARS-CoV-2 Virus
What we know about the new coronavirus and COVID-19