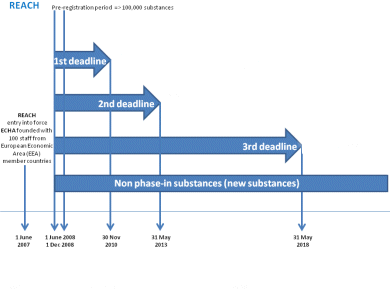
The second deadline of REACH (Registration, Evaluation, Authorization and Restriction of Chemicals) falls on May 31. It applies to chemicals produced in, or imported into, the European Union in annual volumes of 100–1,000 metric tons.
The European Chemicals Agency (ECHA) which manages the technical, scientific and administrative aspects of REACH, expects about 3,000 substances to be registered. Over 4,500 were registered for the first deadline in November 2010 which was looking at substances over 1,000 t. Also the most hazardous substances, e.g., carcinogenic, mutagenic and toxic to reproduction (CMRs) and persistent bioaccumulative and toxic (PBTs) substances, were registered in 2010.
Under normal circumstances, it would be illegal to continue selling affected substances not registered on the EU market after May 31. Companies breaching REACH could face administrative orders, fines, or even criminal charges levied by EU member state enforcement authorities.
However, ECHA said in a statement that it will take into account certain “pressing and exceptional circumstances” in which “registrants may, through no fault of their own, find themselves in difficulty submitting a full registration dossier”.

Image: enlarge
- European Chemicals Agency (ECHA), Helsinki, Finland
- Developments of the registrations
Also of interest:
- REACH – An Overview,
ChemViews Magazine 2012.
DOI: 10.1002/chemv.201200029 - Close to 2nd REACH Deadline – Interview with Christel Musset,
ChemViews Magazine 2013.
DOI: 10.1002/chemv.201300053
Director of Registration at the European Chemicals Agency (ECHA) talks about the benefits and challenges of REACH