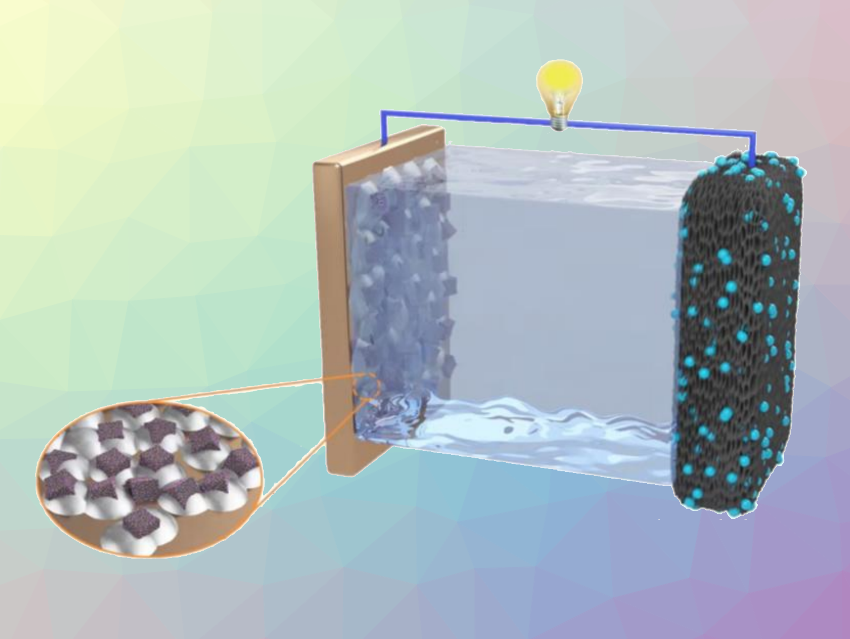
Metallic lithium is a promising anode candidate for next-generation energy-storage systems due to its ultrahigh theoretical capacity. Thus, lithium metal batteries, especially Li–O2 batteries, have attracted research attention. However, metallic lithium anodes present challenges that hamper their practical application, such as volume changes and the formation of lithium dendrites during charge/discharge cycles that can cause dangerous short circuits.
The team first prepared the Zn/Co-ZIF precursor by mixing Co2+ and Zn2+ salts with 2-methylimidazole at room temperature. Then, the precursor was calcined at 800 °C under an argon atmosphere to obtain the desired porous nanocubes. During the calcination, zinc vapor produces pores in the carbon framework that can store lithium and prevent large electrode volume changes. In addition, the nanocubes have many lithiophilic Co- or N-based functional groups, which help with a homogeneous deposition of lithium.
A lithiated composite anode that incorporates the nanocubes shows a more stable voltage profile with a lower overpotential compared with a Li-Cu anode, and the resulting full Li–O2 cell was able to continuously work for more than 200 cycles. This work could be useful for the design of lithium metal anodes for high-performance Li–O2 batteries.
- Constructing bimetallic ZIFs‐derived Zn, Co‐containing N‐doped porous carbon nanocube as the lithiophilic host to stabilize Li metal anodes in Li–O2 batteries,
Huimin Zhao, Huixiang Yin, Ziqi Fu, Zhenzhen Chi, Lin Li, Qingwei Zhang, Ziyang Guo, Lei Wang,
ChemSusChem 2022.
https://doi.org/10.1002/cssc.202200648