Do Stradivarius violins really sound better than all the rest?
This is a difficult question to answer from a scientific point of view, since there is no unambiguous definition for “sound quality”, let alone a definitive relevant measurement process. Sound quality involves a sensory pleasure, the appraisal of which is to a great extent subjective. But without argument only chemistry makes it possible for us to hear the Stradivarius.
7. Determining Sound Quality
7.1 Blind Studies
Going back to the beginning of the 19th century there have been numerous competitive studies trying to pit Stradivarius violins against those of other master violinmakers. The results have consistently been the same: More-skilled and less-skilled jurors failed at picking out the actual Stradivarius.
Probably the most serious comparison was organized in 1977 by the BBC. On that occasion the world famous violinists Isaac Stern and Pinchas Zukerman, together with the distinguished English violin merchant Charles Beare, were challenged to distinguish between the “Chaconne” Stradivarius of 1725, a Guarnerius del Gesu from 1739, a Vuillaume from 1846, and a recently completed instrument from the English master violinmaker Ronald Praill. Stern and Zukerman were allowed to inspect all the violins at their leisure, as well as play them. The violin virtuoso Manoug Parikian then stood behind a curtain and performed on each of them the opening of the Bruch G minor violin concerto, as well as an excerpt from the Chaconne by J. S. Bach. Finally, Stern, Zukerman, and Beare were asked to assign identities to the four violins. The results were sobering, to say the least. Not a single one of the unquestionably competent jurors managed to correctly identify more than two of the four instruments, and two thought the modern violin was the Stradivarius [21].
Comparative tests of this sort are clearly not unproblematic. For example, the player of the instrument generally knows its origin, and may unconsciously express this knowledge in his playing. In a true double-blind test neither player nor listener should be able to see the instrument. Since most musicians in the course of playing find it necessary at least occasionally to glance at the fingerboard, a real double-blind experiment would be extremely difficult to realize.
At the Fourth American Cello Congress, a double-blind test was indeed carried out with 12 celli. Over 140 musicians assembled at the conference, most of them cellists, were asked to assess the sound qualities of six old and six new celli. The one judged the best was in fact an old cello, but those following in places 2–5 were all new instruments. As a group, the new celli were held in higher regard than the old ones.
Methodological objections were raised against this comparison as well: Are 140 musicians, assembled at random at a conference, qualified to judge the sound quality of a collection of celli, or—as in a snide remark of one critic—is this instead a case of “twelve celli being assessed double-blind by 140 double-deaf” listeners?
It is always possible to raise or devise methodological complaints against blind studies involving subjective evaluations. Nevertheless, one is certainly tempted to entertain the notion, after numerous single- and double-blind tests conducted over the course of nearly 200 years, that one is in truth unable to hear any difference between a Stradivarius and instruments from other master violinmakers. This would at least seem to be a serious possibility. In any case, the absolute certainty often associated with the claim that a Stradivarius is without question the finest of all violins—this assertion must surely be regarded as debatable.
7.2 Soloists Opinions
All this notwithstanding, virtually every concert soloist swears by his or her Stradivarius, Guarnerius, or Amati, and holds firmly to the belief that these instruments have better playing qualities, and that they greatly enhance the breadth of their own personal capacities for expressiveness. To quote Anne-Sophie Mutter once again in her characterization of the “Lord Dunn-Raven”: “I love its sound, the beauty and sheer endless depth of its expression, and after more than 20 years I continue to discover new features in this marvelous instrument” [22].
Because each of these violins is itself absolutely unique, and so very valuable, and is associated with such a sonorous name, and because in the past so many master violinists have enjoyed so many great moments on precisely this instrument, the fortunate musician naturally feels a deep respect and esteem for his (or her!) instrument. Consequently he brings to his performance all the skill of which he is capable simply in order to be worthy of the Stradivarius. In a sense he does not want to let it down. The result of this autosuggestion is, for the musician, an optimum performance, both technically and artistically. For the music lover who thereby comes into his own, it is ultimately irrelevant whether it is the Stradivarius, or the violinist, or the two together, or one because of the other that is responsible for creating such a soaring musical peak performance, and thereby for this very special placebo effect.
There actually do exist a very few soloists who refuse to be infected by the Stradivarius myth, and instead swear by instruments produced by modern master violinmakers. Thus, Christian Tetzlaff performs on a violin from a master violinmaker from Bonn, Germany: Stefan-Peter Greiner. He is quoted as having said in an interview: “If I were to subject my Greiner to a double-blind test with a Stradivarius and a Guarnerius, I am certain that no one would be able to say which one was the modern-day instrument. Whenever I perform with an orchestra where the musicians don’t know what sort of instrument I play, I inevitably get asked whether it is a Stradivarius or a Guarnerius.”
8. But Back to Chemistry—Quiet please! The Concert is About to Begin!
The warning signal has already sounded three times. The musicians are tuning their instruments, the conductor and the violinist are walking onto the stage, there are a few last coughs from the audience, and then silence. The virtuoso positions the violin under his chin, the conductor raises his baton, and then there sounds the first violin note: long, swelling, and warm. In the enjoyment of this artistic moment we might easily have ignored altogether the associated chemistry, which—as so often—inconspicuously does us so much good. In this case it makes it possible for us even to hear the Stradivarius. And this through chemistry of the finest sort!
The Strings
The four violin strings may be made of metal, or gut, and/or they could be synthetic. Since the length of each string (l) is stipulated at roughly 33 cm, the corresponding fundamental frequency νo works out to be:
νo= (2 l)–1 (σ/ρ)1/2 = 1.52 (σ/ρ)1/2
The actual pitch (frequency) produced can be altered by the tension on the string σ [kg/(ms2)], as well as by its density ρ [kg/m3], where rather narrow limits are set for the tension: On the one hand a minimum tension must guarantee the precise length of the string, and on the other the tension cannot be too great or else the wooden body of the violin would no longer be able to support it. For this reason the lower strings normally consist of a taut gut or synthetic core, surrounded by metal wire winding as a means of increasing the density ρ. Their precise composition is invariably a closely held trade secret, and even the most elementary mechanical data are never betrayed [24].
This tends to produce highly variable buying habits among musicians: In some cases conservative, with lifelong loyalty to particular manufacturers, and at the other extreme trying every new innovation as soon as it appears on the market.
Nevertheless, every musician must decide whether to use gut or synthetic strings. Strings based on sheep’s intestine (called “catgut”!) consist basically of proteins, which for a while after installation have a tendency to ease off, requiring frequent retuning. Moreover, gut strings are quite sensitive to changes in humidity, since water can be repeatedly incorporated and then released between the extended protein chains [25]. For this reason, a top-quality violin is generally transported in a case in which constant humidity can be maintained. Water incorporation under high humidity conditions may occur not only between protein chains, but also between glucose chains in the cellulose of the wood. This leads to increased damping of the vibrational characteristics of the body of the violin itself, resulting in the sound of the instrument acquiring a dull quality.
Modern synthetic strings are made of nylon, i.e., polyamide chains of the type HN(CH2)6NH–CO(CH2)6CO–)n, which are far less sensitive to incorporation of water, and thus more stable with respect to length. Quoting Anne-Sophie Mutter once again, who swears by synthetic strings (without saying so explicitly): “I have for 20 years used a loosely wound A-string, which offers greater warmth of tone, in some sense more closely resembling the round tone of the catgut-string aesthetic”.
The violin teacher Giovanni Battista Viotti (1755–1824) encapsulated it precisely: “The violin, that is the bow”. Every beginner on the instrument must painfully learn this lesson, because any false motion is punished immediately by generation of a sound that is unpleasant, and indeed to some extent ghastly. Why does the bow make the life of a violinist so difficult? We next delve into the process of tone generation from the standpoint of physical principles.
A string, when fixed at both of its ends, can support only certain specific standing waves, this as a consequence of its defined length. With respect to a particular vibrational frequency ν:
νn = n c (2 l)–1
with n = 1, 2, 3, …
where l is the length of the string, c is the rate of propagation, n = 1 represents the fundamental vibration, and the higher n values correspond to overtones. The characteristic sounds of various musical instruments are a function of relative contributions of various overtones. From this rather simple perspective one can describe to a first approximation the generation of sound in, e.g., a guitar or a piano, but the excitation of a string through use of a bow is a great deal more complicated.
Simply from a visual standpoint, there is no obvious difference in results between using a bow to excite a violin string, or the plucking of a guitar string: In both cases one obtains what amounts to half of a sine wave. But the German physicist Hermann von Helmholtz was able to prove experimentally that here the eye is deceived, since in reality the behavior of a violin string is quite stunning [26]. For a study he published in 1862, Helmholtz developed an ingenious “vibration microscope”. Onto a violin string he glued a white grain of starch, and the objective lens of a microscope was then caused to move back and forth through the action of a tuning fork, in turn stimulated electrically and set perpendicular to the motion of the string. In principle this might be regarded as a crude precursor to the oscilloscope.
In terms of observed vibrational behavior, known here as Helmholtz motion, points on such a bowed string are found actually to follow a triangular path, with the vertex that is located on the string—and falling therefore between the two fixed points (the “kink”, or “Helmholtz corner”)—travelling along the string at the vibrational frequency [33]. With c = λν , and a fixed wavelength of 66 cm, it follows for the usual four strings that the propagation velocities will be: cg = 132; cd = 198; ca = 290 and ce = 436 [m/s].
The naked eye can only perceive the bare outline of this rapid back-and-forth motion—which seems to correspond to half of a sine wave. In other words, it would thus appear to be the case that guitar and violin strings both vibrate in the same way.
The rapid back-and-forth motion of the Helmholtz corner actually has a significant effect on the relationship between motion of a bow and that of the strings (see Fig. 13).
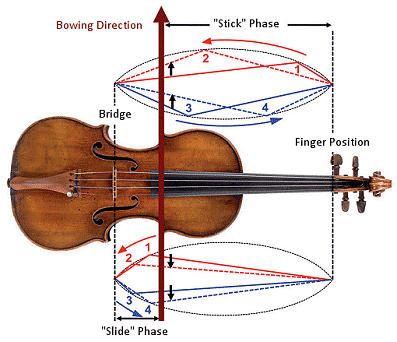
Figure 13. Helmholtz motion of violin strings. In what follows, the bow stroke proceeds upwards (up stroke), that is to say, in the direction from the higher to the lower strings. In the process, the “Helmholtz corner” rotates counter-clockwise. Above: If the Helmholtz corner lies between the bow and the fingerboard, e.g. from 1→ 2 or 3 → 4, both string and bow at their point of contact move in the same direction (heavy black arrow). In this phase, the string should cling as firmly as possible to the bow (the “stick phase”) so that with a minimum application of force a loud and pure tone is achieved. Below: If the Helmholtz corner falls between the bow and the bridge, e.g., from 1→ 2 or 3 → 4, at the point of contact the string and bow move in opposite directions (heavy black arrow). During this phase the string should slide back on the bow in as frictionless a way as possible (the “slide phase”). When the Helmholtz corner then passes the bow in the direction of the fingerboard, the string and bow once again move in parallel, and a new stick-slip cycle can begin. (Violin pictured here: Stradivari’s “Auer”, 1690)
“Proper” bowing technique should by definition cause a string to vibrate as intensely as possible, but with a minimum application of force. This will only be achieved, however, if the pressure of the bow matches precisely the currently applicable “slip-stick” cycle. Learning to meet this objective entails a great deal of practice!
Consider a bit more closely what is actually occurring at the point of contact between bow and string. On the bow itself, some 120–150 individual horse hairs are fastened such that they can be moved, as a group, across the strings. Friction causes the string in question initially to follow the bow, until the restoring force of the string begins to exceed the relevant frictional force, at which point the string springs back away from the bow hairs—until, that is, the frictional force again exceeds the restoring force, leading to onset of a new slip-stick cycle.
If a violinist with a newly strung bow were to immediately begin playing, the resulting sound would be horribly disappointing: No full tone whatsoever, only a pathetic chirping and squealing. The reason is complete lack of synchronization between slip-stick cycles and cyclical rotation of the “kink” or Helmholtz corner. If string instruments always sounded like this, no composer would ever have written a violin concerto, and there would be no string section in a symphony orchestra. We would in effect be condemned to listening exclusively to the brass and woodwinds.
So what is to be done? Synchronization of the slip-stick cycle with rotation of the Helmholtz corner is achieved only if the string remains firmly fixed to the bow hairs during the “stick” phase, and then slides back without friction during the “slip” phase. Shifting of the adhesion from 100 % to 0 % and then back again—and this perhaps 100 times per second—sounds like something that would require a miracle. Accomplishing miracles like this requires the intervention of chemistry, which here entails rubbing a bit of rosin also known as “colophony” on the bow hairs. It’s important to be emphatic regarding this point: Even a Stradivarius sounds miserable in the absence of rosin. Only with rosin is it possible for any violin to produce the treasured tones assuming, of course, that it is also played skillfully—but that’s another story altogether …
8.3 Rosin (Colophony)
The term “colophony” comes from the name of the ancient Greek city Colophon, which was situated between Smyrna and Ephesus (the site of present-day Izmir, Turkey). Even in antiquity it was known for its conifers, which were a prime source of valuable resin. Today our colophony is derived from the resin of the North American longleaf pine (Pinus palustris) or the European Scotch pine (Pinus sylvestris). Its collection involves use of a plane to make a V-shaped cut into the bark, one deep enough to reach the wood responsible for conducting sap, and then gathering the resultant tree gum or resin (pine balsam) (see Fig. 14).

Figure 14. The collection of rosin (colophony). Photos: Herbert Kohlross/www.schwarzfoehre.at.
More generally, when the bark of a pine tree is breached, a resin (balsam) is secreted which, after distillation of ca. 20 % volatile turpentine, leaves behind a glassy, pale yellow to red-brownish solid: the colophony. This material softens in the range 60–70 °C, and consists largely of a mixture of isomeric resin acids (see Fig. 15) [27, 35].
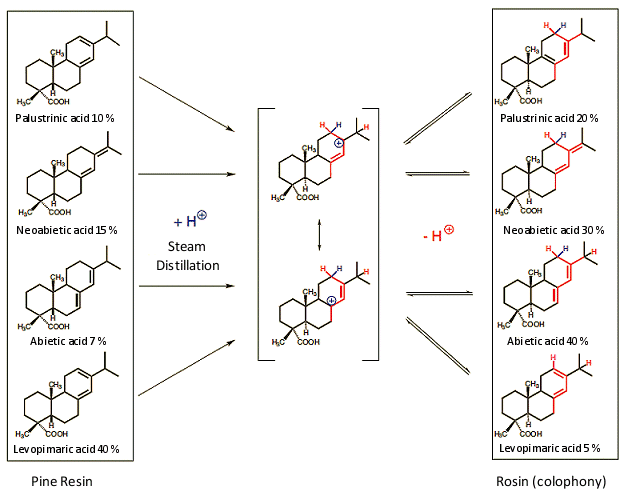
Figure 15. Chemical constituents of rosin.
Apart from these resin acids, however, the colophony also includes non-volatile resin-acid derivatives, such as alcohols and keto compounds. The resin acids belong to the category of tricyclic diterpenes, whose basic structures result from the coupling of four C5-isoprene units (isoprene = 2-methyl-1,3-butadiene).
In the course of steam distillation, levopimaric acid in particular rearranges to abietic acid, and the latter is in fact often prepared in this way [28]. Mechanistically, the rearrangement entails protonation at a terminal carbon atom of the conjugated double bond system, leading to a resonance-stabilized carbocation. The latter, through proton-transfer to the neighboring (allylic) CH2-group, generates a rearranged diene. The product ratio in the resulting equilibrium mixture is determined by the relative stabilities of the different conjugated dienes. Only after this rearrangement does the rosin show the physical properties so prized by violinists.
Actually, high-quality violin rosin involves even more preparation, this time the selective addition of other natural resins from fir trees, spruce, and larches in order to achieve just the right softening point. Those instruments with heavier strings like celli and basses require a somewhat softer rosin. The precise composition of first-rate violin rosin will be based on a time-honored formulation, typically held as a closely guarded trade secret.
How is it that rosin has such a dramatic influence on the sound of a violin?
Before a newly restrung bow is used for the first time, the hairs are stroked 10–12 times with rosin (6–7 strokes suffice for a bow that has already undergone a rosin treatment). The newly applied rosin adheres to the surface of the hairs, and the added stickiness results during bowing in increased static friction, at least so long as the Helmholtz corner is moving between the bow and the fingers. Should the Helmholtz corner pass beyond the bow, in the direction of the bridge, the corner’s motion is reversed, and the string begins to pull along the bow hairs in the opposite direction. This causes heat to be released, the rosin melts, and the string slides along a rosin film as if on a waterslide (Fig. 16).
Upon its renewed passage under the bow, the Helmholtz corner again travels with the bow, friction decreases, the rosin again solidifies, the string and the bow hairs fuse together once more, and the bow again exerts a strong pull on the string. This cycle of melting-resolidifying repeats itself several hundred times per second.
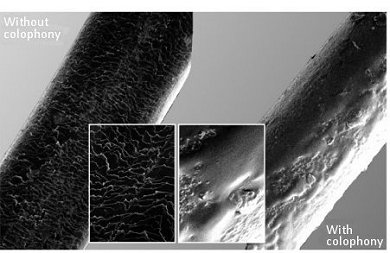
Figure 16. Bow hairs before and after treatment with rosin (colophony). The surface of a horse hair is by its nature somewhat scaly, but still nevertheless rather smooth, just as is human hair (left). After rosin has been rubbed in, however, and right away it is used for playing on a violin, it is plainly evident (right) that the rosin layer on the surface of the hair has already undergone a process of melting and resolidification [29].
9. In Summary …
Moments of musical glory will be experienced only in the fortunate case that a great many details fall properly together: A wonderful composition is skillfully interpreted by a musician in a good mood in front of an attentive audience, in a room with good acoustics, and on an instrument built by a master. The audience will be completely oblivious of the fact that only thanks to chemistry is a reasonable level of listening pleasure made possible with respect to a string instrument. Starting with rosin for effective transfer to the strings of the motion of the bow, through the vibrational characteristics of strings that have a synthetic core, to the priming and varnishing of the instrument, which contributes significantly to both beauty and sound—not only of a Stradivarius, but to every other violin as well.
If you would like to have a chance yourself to play on a Stradivarius, ChemistryViews magazine will be glad to help you out! You’ll need only to bring along your talent and many years of experience with one or more good violin teachers, together with at least 5–6 hours of daily practice. Then you should obtain an application form from, for example, the Nippon Musical Society at www.nmf.or.jp/english/ fill it out, and send it in [30]. If after that you succeed in mesmerizing Lorin Maazel and his fellow referees, nothing will be standing in the way of your being able to play on a genuine Stradivarius! We are already crossing our fingers for you here ….
Then, when you enrapture your audience with the first note, listeners with a chemical background will appreciate the sound even more. Is it not wonderful that only because of a synthetic isomerization of natural resin acids is it possible for the violinist to express his or her artistic interpretation through the bow via the—again synthetic—core of the strings to the body of the instrument, and from there to the ears of the listener? There’s no way to get around the fact: Only through chemistry have such transcendental pleasures become possible!
References
[21] A. Coggins, The Strad 2007, February, 52, also to be found under http://www.abcviolins.com/blind-listening-tests
[22] A.-S. Mutter, private communication, 2009.
[23] loc. cit. The Strad 2005, July.
[24] N. C. Pickering, J. Catgut Acoust. Soc. 1985, 44, 6.
[25] D. G. Hunt et al., Nature 1996, 379, 681. DOI: 10.1038/379681a0
[26] G. Weinreich, Am. J. Phys. 1993, 61, 1067. DOI: 10.1119/1.17353
[27] W. Lange et al., Holz als Roh-und Werkstoff. 1987, 45, 345. DOI: 10.1007/BF02605851
[28] Org. Synth. 1963, Coll. Vol. 4, 1. Link
[29] H. Pöcherstorfer, wissenschaftliche Hausarbeit, Institut für Wiener Klangstil, http://iwk.mdw.ac.at/Forschung/deutsch/strinst/Bogenh/bogenh.htm
[30] Other foundations and organizations: Antonio Stradivari Cremona, Stiftung Habisreutinger, Regierungsgebäude, 9001 St. Gallen, Switzerland; The Stradivari Society, 410 South Michigan Avenue • Suite 1014 • Chicago, IL 60605, USA.
Aknowledgement
I wish to thank the following colleagues for their assistance in preparing me in this musically, physically, and chemically complex field: Specifically, Fabian Adam, Berlin, Germany, for my first (and last!) violin lesson, and Dr. Andreas Schäfer, Free University of Berlin, for his patience with the Wolf tone produced when the played note matches the resonance frequency of the violin and likened to a wolf’s howl. I am grateful to Dr. S. Streller and Dr. P. Winchester, Free University of Berlin, for help in completion of the manuscript. To Dr. Francis Schwarze, EMBO Basel, Switzerland, Professor M. Bertsch, Vienna, Austria, Dr. H. Steger, Polytec, Waldbronn, Germany, Drs. C. Y. Barlow and J. Woodhouse, University of Cambridge, UK, Dr. H. D. Grissino-Mayer, University of Tennessee, Knoxville, USA, and the Smithsonian Institution, Washington, D. C., USA, I am grateful for reprint permissions and for patience in the clarification of technical issues. My thanks go also to Ruggero Noto La Diega for his linguistic help in interpreting the Stradivari letter. Finally, I am deeply indebted to Anne-Sophie Mutter for the description she provided of her personal sensations with respect to her “Lord Dunn-Raven“ Stradivarius.
Author
Prof. Klaus Roth, Freie Universität Berlin, Germany.
The article has been published in German in:
and was translated by W. E. Russey.
Chemical Secrets of the Violin Virtuosi — Part 1
What made Stradivari’s violins so special? Klaus Roth looks at the important role of chemistry in Stradivari’s workshop and instruments
Chemical Secrets of the Violin Virtuosi — Part 2
In part 2 we consider from a chemical point of view those components of the Stradivarious that are the principal sources of sound characteristics.
Other articles by Klaus Roth published by ChemVistryiews magazine:
- In Espresso — A Three-Step Preparation
Klaus Roth proves that no culinary masterpiece can be achieved without a basic knowledge of chemistry
DOI: 10.1002/chemv.201000003 - In Chocolate — The Noblest Polymorphism
Klaus Roth proves only chemistry is able to produce such a celestial pleasure
DOI: 10.1002/chemv.201000021 - In Sparkling Wine, Champagne & Co
Klaus Roth shows that only chemistry can be this tingling
DOI: 10.1002/chemv.201000047 - In Chemistry of a Hangover — Alcohol and its Consequences
Klaus Roth asks how can a tiny molecule like ethanol be at the root of so much human misery?
DOI: 10.1002/chemv.201000074 - In The Chemist’s Fear of the Fugu
Klaus Roth shows that the chemist’s fear of the fugu or pufferfish extends as far as the distinctive and intriguing poison it carries
DOI: 10.1002/chemv.201000104 - In Chemistry of a Christmas Candle
Klaus Roth explains that when we light a candle, the chemistry we are pursuing is not only especially beautiful, but also especially complex
DOI: 10.1002/chemv.201000133 - In Pesto — Mediterranean Biochemistry
Klaus Roth uncovers the nature of this culinary-chemical marvel, and thereby comes to enjoy it all the more
DOI: 10.1002/chemv.201200001 - In Boiled Eggs: Soft and Hard
Klaus Roth examine an egg on its journey from hen to table to ensure the perfect breakfast egg
DOI: 10.1002/chemv.201200018 - Video Interview with Klaus Roth
See all articles published by Klaus Roth in ChemVistryiews Magazine


