The periodic table of the elements is one of mankind’s greatest discoveries in nature since it encompasses all the building blocks that bind our universe together at its heart: from the tiniest virus to the most distant galaxy. Last time, we discussed the difficult road towards element 118. In this part, we have a look at the first synthesis of element 118, its properties, and how new elements are named.
5. First Actual Synthesis of Element 118
In 2002, a research group consisting of scientists from the Joint Institute for Nuclear Research in Dubna, Russia, together with the Lawrence Livermore National Laboratory in Berkeley, CA, USA, commenced with the first synthetic effort to produce element 118 by bombardment of californium-249 with calcium-48 ions [21–23]. Calcium-48, with a natural abundance of only 0.19 %, is very rare, and correspondingly costly (USD 200,000/g). For a light element (Z = 20), it is extraordinarily neutron-rich with a neutron count of 28, and for that reason, especially well-suited to the synthesis of stable, heavy nuclei.
For 100 days, the team bombarded a target consisting of 10 mg of 249Cf (0.23 mg/cm2) with a calcium-48 beam of 2·1012 roughly 17-fold positively-charged ions per second; in the course of three months of irradiation, a total of 2·1019 calcium ions. During this entire period, they obtained evidence for only a single decay sequence attributable to element 118!
4820Ca +24998Cf → 297[118]
Work was resumed in 2006, revealing, as required, two additional relevant decay sequences [24] (see Fig. 3). This time, numerous prerequisites had to be fulfilled for recognition of the discovery, published in detail by the International Union of Pure and Applied Chemistry (IUPAC) and the International Union of Pure and Applied Physics (IUPAP) [25, 26]. Sufficient decay series consistent with element 118 were indeed established, but none of the isotopes involved were previously known. There was, thus, a failure to create a link to known isotopes, so recognition had to be denied [27]: “The three analyzed decay series for an isotope of element Z = 118 are in good mutual agreement, but in the absence of anchoring to known nuclei, the requisite criteria for recognition are not fulfilled.”
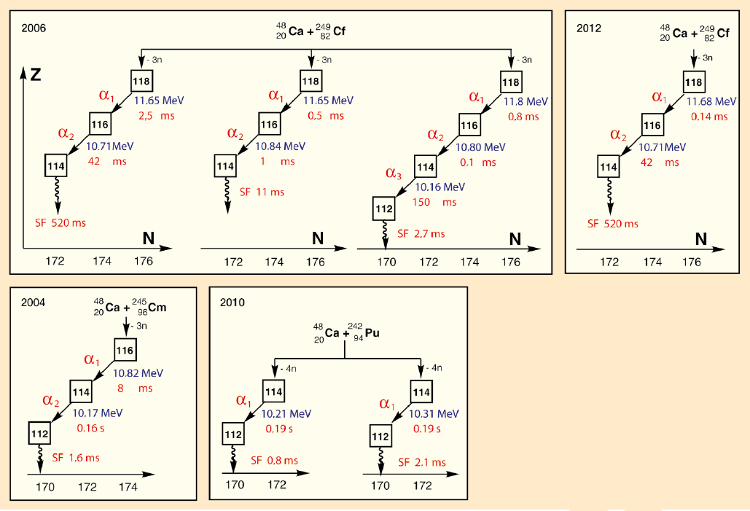 |
|
Figure 3. Experiments relevant to the discovery of element 118. |
It was possible, however, to confirm the decay series for element 118 via independent studies involving lateral entries. Thus, elements 116 and 114 were prepared via independent paths by bombardment of curium and plutonium, respectively, with calcium-48 ions, and their decay series were determined (see Fig. 3). These decay series were found to be consistent with the corresponding parts of the decay series for element 118. As a consequence, IUPAC/IUPAP indeed released a positive judgment:
“The 2006 Dubna–Livermore collaboration of Oganessian et al. produced three concordant decay chains commencing with 294118. This result was confirmed in 2012. Three other independent heavy element fusion studies served to identify and confirm the existence and decay properties of 294118 descendants 290Lv and 286Fl serving to link atomic numbers through cross bombardments. The Dubna–Livermore 2006 collaboration has satisfied the criteria for discovery and its claim is now acknowledged as validated.”
Thus, it was officially recognized that the Russian–American research group under the direction of Yuri Oganessian had indeed discovered element 118. The President of the IUPAC Inorganic Division then requested an appropriate suggestion for a name and symbol for the new element. Consistent with additional regulations, the plenary session of IUPAC voted on the acknowledgment of the discovery and the naming of element 118.
6. The Naming of the Elements 113–118
The discoverers of an element have the right to suggest a name for it, but they do not have a completely free choice. IUPAC and IUPAP have developed certain applicable rules [28, 29], and the name of the new element is to be derived from:
- a) a mythological concept or character, including astronomical objects
- b) a mineral or similar material
- c) a place or geographical region
- d) a characteristic of the element
- e) the name of a scientist
In the interest of standardization, suggested element names for certain elements should always have one of the following endings:
- Groups 1–16, including elements of the f-block: “-ium”
- Group 17 (halogens): “-ine”
- Group 18 (noble gases): “-on”
Decisions regarding element names are difficult for all the parties involved: not just the discoverer, but also IUPAC/IUPAP. Discoverers, with their own egos, necessarily live in a specific complex political environment, and IUPAC/IUPAP and their international experts also do not exist in a vacuum. In the Cold War era, this led to disputes that have sometimes been quite grotesque. Thus, over a period of three decades, element 104 was identified in schoolbooks in the United States as rutherfordium, but in Russian as kurchatovium. Fortunately (hopefully?), this period is over. Today, the synthesis of the heavy transactinides shows how advantageous it is when research groups from different countries apply their expertise to common projects.
6.1. Element 113: Nihon (Nh)
Element 113 was first prepared—after years of effort—by a Japanese group at RIKEN led by Kosuke Morita. The suggested name and symbol were designed to honor their homeland: “Nihon” (land of the rising sun), with the accompanying symbol Nh.
Morita and his team began bombarding a bismuth target with zinc ions in 2003, and by April 2005, they had detected two consistent decay series for element 113. This was not considered sufficient for recognition, however. Only in August 2012, after seven additional years of irradiation, was the long-yearned-for third decay series observed. With their endless patience and tenacity, the research group showed the limit of today’s technical possibilities: one decay series in two years!
Preparation of the three other elements recognized in 2017—numbers 115, 117, and 118—was the result of a close collaboration between Russian (Dubna) and US (Berkeley) scientists. This had the further advantage that the parties involved were able to agree on an amicable compromise with regard to the matter of naming in the preliminary stages.
6.2. Element 115 – Moscovium (Mc)
The Russian capital was honored with the name moscovium (Mc), just as had already been the case for the research site Dubna, only 100 km away from Moscow, with element 105 (dubnium).
6.3. Element 117 – Tennessine (Ts)
The ending “ine” reveals that this element is associated with the 17th group in the periodic table: the halogens (fluorine, chlorine, bromine, etc.). The suggested name was surprising for many, since element 117’s discovery was attributed to the Dubna/Berkeley team of Yuri Oganessian. But a closer look at the synthesis explains the background:
4820Ca +24997Bk → 293[117] + 4n
The bombardment of berkelium-249 with calcium-48 ions had been planned by Oganessian’s team for a long time. The problem was that there was only one place on Earth where a few milligrams of the required berkelium might be prepared: in the high-flux reactor at the Oak Ridge National Laboratory (ORNL) in the state of Tennessee, USA [30, 31]. There one could actually buy24997Bk, albeit at a price of USD 185 per microgram (!), not including packaging. The planned experiment required 20 mg. It was necessary to take into account not only the high cost, but also the short half-life of24997Bk: only 330 days. A comprehensive American-Russian masterplan was developed for the preparation of element 117, covering all aspects of the timing of the experiment itself, as well as further processing and transport over thousands of kilometers.
In the spring of 2008, 40 g of curium-244 was introduced into the ORNL high-flux reactor, and subjected to extremely high neutron irradiation for 23 days. After the fuel was exhausted, it was replaced, and the curium sample was irradiated for another 23 days. Following eleven such cycles over a total of 250 days, 22 mg of 24997Bk had been prepared, and over the next six months, it was isolated from the curium sample and then purified (see Fig. 4).
 |
|
Figure 4. The starting material for the tennessine synthesis: berkelium-249. |
A solution of the resulting berkelium chloride was placed in five lead receptacles and transported to Moscow on a commercial flight. The collaborative efforts between the researchers had proceeded without problems, however, the same could not be said with respect to subsequent sample transport. Twice, the berkelium containers were refused at the Russian border because of missing or incomplete documents, and then sent back to New York. Only on the third try did they reach the Russian Research Institute for Atomic Reactors in Dimitrovgrad, where the target discs were prepared. Finally, the actual experiment could begin in Dubna on July 27, 2009. A first decay series of element 117 was found on August 20, 2009, and over the course of the next six months, five additional decay series were recorded.
6.4. Element 118 – Oganesson (Og)
The ending “-on” implies that this element is recognized as one of the “noble gases” (group 18). The name assigned to it is a tribute to Yuri Oganessian (see Fig. 5), who became only the second living scientist for whom an element was named after Glenn T. Seaborg with seaborgium. When asked what his feelings were about that, Oganessian replied [23]:
“For me, it is an honor. Discovery of element 118 was achieved by scientists from the Joint Institute for Nuclear Research in Russia and the Lawrence Livermore National Laboratory in the United States, and it was my colleagues who suggested the name oganesson. My children and grandchildren have been living in the United for decades already, but my daughter wrote to me when she heard about it that she was unable to sleep that night because she was crying so much. My grandchildren, in contrast, like all young people, barely reacted.”
 |
|
Figure 5. Yuri Oganessian on an Armenian stamp. |
For chemists, the question arises whether oganesson is not only formally a noble gas, but whether it also behaves physically and chemically like one. Many chemists remain skeptical about quantumchemical calculations, especially with respect to the transactinides, since the high velocities of their inner electrons (70 % of the speed of light in the case of copernicium, Z = 112) are more difficult to take into account (relativistic effects). But if Dmitri Mendeleev was able to make certain precise and correct predictions even in the 19th century using only pencil and paper, we should perhaps place more trust in our theoreticians. In any case, based on their calculations, the opening question “Is element 118 a noble gas?” can quickly be answered by “Oganesson is guaranteed not to be a noble gas, but perhaps rather a “noble liquid”, with a boiling point of 50–110 °C [32].
Moreover, oganesson should react with fluorine to give the stable compounds OgF2 and OgF4 [33, 34], where OgF4 would not be planar like xenon tetrafluoride, but tetrahedral. This cannot yet be verified experimentally, since the four synthetic oganesson atoms were gone after a single millisecond. Let us, therefore, look toward more stable oganesson isotopes, and prepare to be surprised by their chemistry.
7. How Do Things Proceed from Here?
With respect to the synthesis of element 119, nuclear researchers are still at the starting gates. Hideto En’yo from the Japanese research institute RIKEN has announced a bombardment of curium with vanadium ions
96Cm + 23V → [119]
and the Oganessian group wants to fire titanium ions at berkelium
97Bk + 22Ti → [119]
It is nearly certain that these experiments will bump up against the limits of current technology. The synthesis of yet heavier elements certainly presupposes additional major technical developments. We will have to see over what time period the necessary progress will be achieved. We, therefore, wish our nuclear scientists both good ideas and good luck in their further search for new elements. However, not too much luck, since if they were suddenly to stumble across stable isotopes, no one would even notice it.
We mustn’t forget that the heavy nuclei are so far recognizable only on the basis of their radioactive tracks. So if stable nuclei were to emerge, we wish nuclear scientists simultaneously the technical possibility of preparing weighable amounts of the corresponding elements. Then one might be able to study chemical reactions of their electron shells. And that would be exciting since, for example, theoretical computations suggest that element 123 should possess three partially-filled orbitals in three different shells (8s2 8p 7d 6f). We should be especially anxious to study element 125, because it will involve occupied g-orbitals for the first time ([Og] 8s2 8p 6f3 5g). Merely the forms of the various 5g-orbitals cause a desire to examine their chemistry (see Fig. 6). We will wait expectantly!
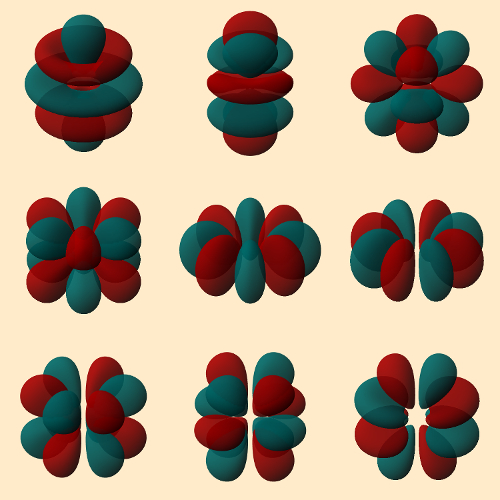 |
|
Figure 6. The 5g orbitals; from left to right and from top to bottom [35]: |
References
[21] Y. T. Oganessian et al., Results from the First 249Cf+48Ca Experiment, Lawrence Livermore National Laboratory Report 2003.
[22] Y. T. Oganessian, Superheavy elements, Pure. Appl. Chem. 2004, 76, 1715. https://doi.org/10.1351/pac200476091715
[23] R. Gray, Mr Element 118: The only living person on the periodic table, New Scientist 2017, April 15, 40.
[24] Y. T. Oganessian et al., Synthesis of the isotopes of elements 118 and 116 in the 249Cf and 245Cm+48Ca fusion reactions, Phys. Rev. C 2006, 74, 044602. https://doi.org/10.1103/PhysRevC.74.044602
[25] A. H. Wapstra, Criteria that must be satisfied for the discovery of a new chemical element to be recognized, Pure Appl. Chem. 1991, 63, 879. https://doi.org/10.1351/pac199163060879
[26] R. C. Barber et al., Discovery of the transfermium elements, Pure Appl. Chem. 1993, 65, 1757. https://doi.org/10.1351/pac199365081757
[27] P. J. Karol et al., Discovery of the element with atomic number Z = 118 completing the 7th row of the periodic table, Pure Appl. Chem. 2016, 88, 155. https://doi.org/10.1515/pac-2015-0501
[28] W. H. Koppenol, Naming of new elements (IUPAC Recommendations 2002), Pure Appl. Chem. 2002, 74, 787. https://doi.org/10.1351/pac200274050787
[29] W. H. Koppenol et al., How to name new chemical elements (IUPAC Recommendations 2016), Pure Appl. Chem. 2016, 88, 401. https://doi.org/10.1515/pac-2015-0802
[30] J. S. Bardi, An Atom At The End Of The Material World, Inside Science 2010, April 8.
[31] K. Chapman, What it takes to make a new element, ChemistryWorld 2017, January 22.
[32] C. S. Nash, Atomic and Molecular Properties of Elements 112, 114, and 118, J. Phys. Chem. A 2005, 109, 3493. https://doi.org/10.1021/jp050736o
[33] Y.-K. Han, Y. S. Lee, Structures of RgFn (Rg = Xe, Rn, and Element 118. n = 2, 4.) Calculated by Two-component Spin−Orbit Methods. A Spin−Orbit Induced Isomer of (118)F4, J. Phys. Chem. A 1999, 103, 1104. https://doi.org/10.1021/jp983665k
[34] K. S. Pitzer, Fluorides of radon and element 118, J. Chem. Soc. Chem. Comm. 1975, 760. https://doi.org/10.1039/C3975000760B
[35] M. Winter, The Orbitron, winter.group.shef.ac.uk.
The article has been published in German as:
- Ist das Element 118 ein Edelgas?,
Klaus Roth,
Chem. unserer Zeit 2017, 51, 418–426.
https://doi.org/10.1002/ciuz.201700838
and was translated by W. E. Russey.
New Kids on the Table: Is Element 118 a Noble Gas? – Part 1
The synthesis of heavy elements
New Kids on the Table: Is Element 118 a Noble Gas? – Part 2
The difficult road towards element 118
New Kids on the Table: Is Element 118 a Noble Gas? – Part 3
The first synthesis of element 118, its properties, and naming new elements
See similar articles by Klaus Roth published in ChemistryViews Magazine




