Prussian blue was the discovery of the century in 1706. Many legends and stories swirled around this pigment and its very lucrative and initially secret production process. In the end, the process was revealed, but it took over 100 years to understand the pigment and only new chemical history research allows us to close the case.
10 The Genesis of Prussian Blue in Detail
Let us try to gather an overall picture of the information available today about the production of Prussian blue in 1706 (see Fig. 16). Our sources are:
- Dippel and Diesbach’s chemical activities (1706)
- The leaked and published Prussian Blue synthesis (1724)
- Dippel’s memories as recorded by Senckenberg (1732)
- Our modern interpretation of the chemical processes, supplemented with some experiments
 |
|
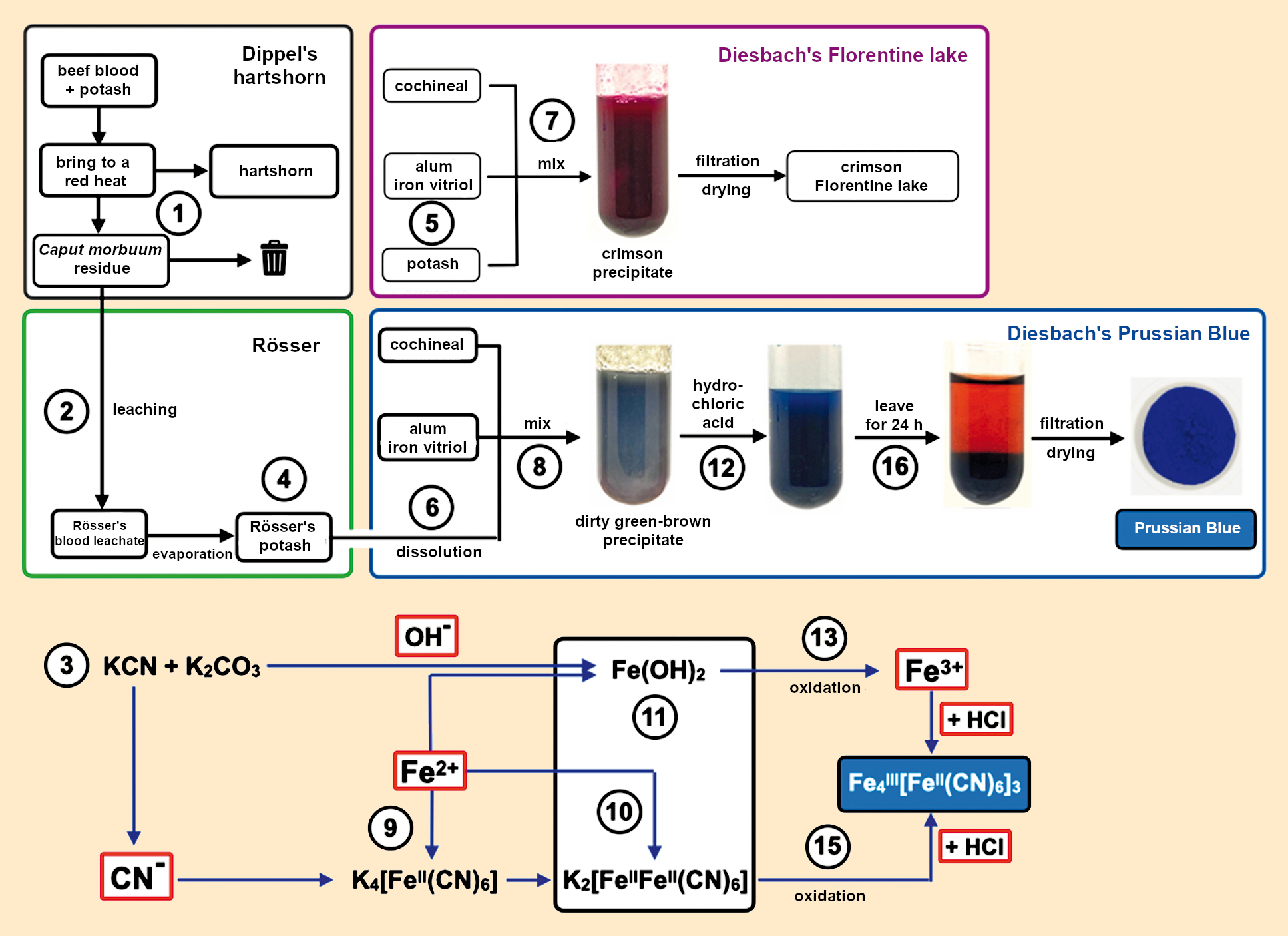
Figure 16. The first synthesis of Prussian blue from 1706. |
10.1 Sequence of Events to the Discovery of Prussian Blue
Based on these sources, the following sequence of events can be reconstructed for the discovery of Prussian blue in the year 1706:
- The discovery of Prussian blue began in 1706 with Dippel’s production of a large amount of hartshorn. When the starting materials, dried beef blood and potash, were calcinated together, about six pounds of residue were formed (1). Having been brought to a red heat, this residue was unusable to the alchemist’s mind and was thrown out. Since the early 19th century, we have known that such residues contain potassium cyanide.
- Against Dippel’s advice, Rösser extracted (leached) this residue out with water (2). The filtrate (Rösser’s blood leachate) was a potash solution containing potassium cyanide (3).
- After evaporation, a white solid mixture of potash and potassium cyanide (Rösser’s potash) was obtained (4). At this point, Rösser made a momentous error in mistaking the white powder for pure potash and correspondingly mislabeling its container.
- Sometime later, Lieutenant Diesbach wanted to produce purple Florentine lake, as he had many times before. For this, he needed some potash (5).
- Because he was out of potash, he used some of the potash mislabeled by Rösser (6).
- Diesbach must have been puzzled when he mixed all of the ingredients and obtained a dirty, green-brown precipitate (8) instead of the usual purple precipitate (7).
- The purple precipitate obtained from the established method of producing Florentine lake (7) was formed after the addition of strongly basic potash solution to a weakly acidic solution of cochineal pigment, iron vitriol (iron(II) sulfate, FeSO4), and alum (potassium aluminum sulfate, KAl(SO4)2). Because of the excess potash, the alkaline solution produced iron(II) hydroxide (green) and aluminum hydroxide (white) precipitates. The cochineal pigment reacted with iron and aluminum ions to form colored complexes that were adsorbed onto the aluminum hydroxide precipitate.
- The precipitate (8) had a different composition from this usual product. Upon mixing, the cyanide and iron ions immediately react to form hexacyanidoferrate anions (9, yellow blood leachate salts). Some of the excess iron(II) ions react with hexacyanidoferrate anions to produce insoluble Berlin white (10), any remaining excess metal cations precipitated out as hydroxides: aluminum as white Al(OH)3 and iron(II) as dirty green iron(II) hydroxide (11). In this case, the iron and aluminum complexes of the cochineal pigment were also adsorbed onto the precipitates.
- The pigment produced by the cochineal beetles also binds to the iron oxide and aluminum oxide precipitates.
- Rösser tried to save what he could by adding hydrochloric acid to the mixture (12). When making Florentine lake, this always led to deepening of the color because the substrate (aluminum hydroxide) would partially dissolve.
- To his surprise, this time the addition of the hydrochloric acid caused the red to disappear entirely, and the first Prussian blue was made (12). The transition from an alkaline to an acidic medium caused the metal hydroxides to dissolve, forming aluminum and iron(II) ions. Because all of the reactions occurred in the presence of air, the iron(II) ions were slowly oxidized to iron(III) ions under both the alkaline and acidic conditions (13). In the acidic solution, these ions immediately reacted with the yellow blood leachate salts (9) to make Prussian blue (14). The Berlin white was also slowly oxidized to Prussian blue by oxygen in the air (15), causing the blue color to intensify over the next 24 hours.
- Within hours, the completely insoluble Prussian blue precipitated out (16), and it was clear that the supernatant solution contained dissolved red cochineal pigment. Simple filtration and drying yielded the magnificent blue pigment (17).
10.2 Coincidence Becomes Luck When it Encounters Talent
In recent years it has been possible to significantly clear up the details of the discovery of Prussian blue in 1706. It was not planned and was not a stroke of genius or divine inspiration. Instead, it was an accumulation of independent coincidences. In hindsight, it is clear to us that the chymists got very lucky. However, this seems slightly disparaging, which they do not deserve because they didn’t just wonder at the coincidence but investigated it and used it as a springboard. Succinctly put: “Coincidence becomes luck when it encounters talent” [35].
At the time, the chymists summarized the surprising formation of Prussian blue at the end of their production process as follows:
The blood transfers its “colored material” to the potash during calcination, the blood leachate obtained consists of potash and “colored material” and the blood leachate transfers the “colored material” to the iron, giving it this magnificent color [19].
Today we would sum up the surprising formation of Prussian blue at the end of the production process of the time as follows:
During the calcination, the blood transfers its nitrogen to the potash as cyanic acid, the blood leachate consists of potash and potassium cyanide and transfers the cyanide to the iron, giving it this magnificent color.
Of course, today, after 300 years of research, we have a better understanding of the chemical reactions that led to the discovery of Prussian blue. However, this comparison shows that we aren’t so superior. On the contrary: Kudos to the chymists!
Acknowledgments
My thanks go to Dr. Alexander Kraft, Eichwalde, Germany, for his support in the incorporation of the chemical historical details. This article is based on his research results. Professor Ulrich Abram, Dr. Hans Bauer, Professor Hans Hartl, Tobias Hirsch, Dr. Andreas Schäfer, Dr. Sabine Streller, Berlin, Dr. Gustav Penzlin, Bonn, Dr. Achim Lunk, Markkleeberg, and Professor Jost Weyer, Hamburg, Germany, provided advice.
The detailed pharmacy historical look at the portrait of Caspar Neumann was made available to us by pharmacist Günter Bergmann, Wallgau, Germany [21]. Permission to print was given by OTL Dr. T. Müller and OL S. Höpfl at the Bundeswehr Medical Academy, Munich, Germany, and unbureaucratic access to the original painting under COVID conditions was made possible by Johanna Stapelfeldt, curator of the Humboldt Laboratory in the Humboldt Forum, Berlin, Germany. Special thanks to Dr. Sabine Streller and Professor Claus Bolte for my guest stay in their research group’s laboratory.
References
[35] H.-J. Quadbeck-Seeger, Der Wechsel allein ist das Beständige, Wiley-VCH Verlag, Weinheim, Germany, 2002.
The article has been published in German as:
- Berliner Blau – Entdecker und Verräter,
Klaus Roth,
Chem. unserer Zeit 2021, 56, 34–49.
https://doi.org/10.1002/ciuz.202100033
and was translated by Caroll Pohl-Ferry.
Prussian Blue: Discovery and Betrayal – Part 1
Around 1700, Berlin was a colorful center of innovation – a scene that led to the discovery of the century: Prussian blue
Prussian Blue: Discovery and Betrayal – Part 2
How Johann Konrad Dippel almost ruined the Prussian blue business
Prussian Blue: Discovery and Betrayal – Part 3
Who leaked the secret recipe for Prussian blue?
Prussian Blue: Discovery and Betrayal – Part 4
How was Prussian blue really discovered—with luck or a stroke of genius?
The Author
Professor Klaus Roth from the Free University of Berlin, Germany, is a regular author for ChemistryViews.
See similar articles by Klaus Roth published on ChemistryViews.org