Asparagus (Asparagus officinalis) is highly prized in culinary circles (see Fig. 1). Every April, Germans, in particular, impatiently await the first asparagus. It is then eaten in large amounts until June 24, when the asparagus season ends—120,000 tons in 2017.
However, enjoying asparagus has an unpleasant side-effect that sets it apart from other vegetables, but is seldom talked about: It makes urine stink. Benjamin Franklin (1706–1790) described this in drastic terms: “A few stems of asparagus eaten, shall give our urine a disagreeable odor.” This sounds like exciting chemistry!
.jpg) |
|
Figure 1. Asparagus (Asparagus officianalis). White asparagus (left) grows underground. It is picked as soon as its tip reaches light (center). Green asparagus (right) is not grown in a hill of soil but is exposed to the sun. It therefore produces the substances needed for photosynthesis, which make it green. |
1. The First Whiff
1.1. Asparagine
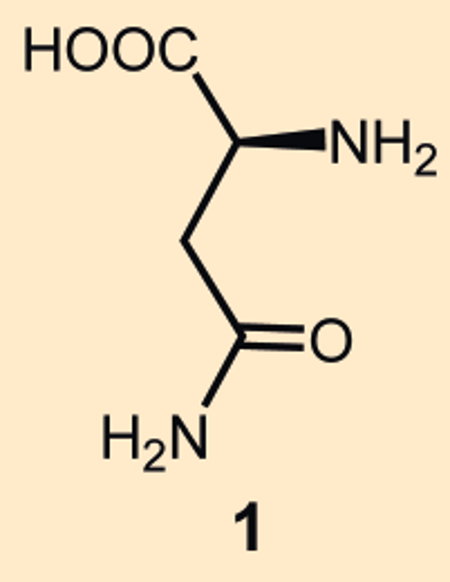
The question of what substances are behind the unpleasant odor of asparagus urine was taken up by Auguste-Arthur Plisson (†1832) and Étienne-Ossin Henry (1798–1873) as early as 1830 [1]. They studied asparagine (1), an amino acid isolated from asparagus only a short time before (see Fig. 2) [2,2a], assuming it was a precursor to the odor. In an experiment on themselves they ingested “very considerable” quantities of asparagine, but the typical urine odor was not detected. Asparagine (1) was, thus, eliminated as the source of the smell.
 |
|
Figure 2. Asparagine. |
1.2. Methyl Mercaptan
Albert Hilger (1839–1905) made a noteworthy attempt to isolate the odorous material in 1874. The description of the experiment in his paper “On Abnormal Components of Urine after Enjoying Asparagus Shoots” is so precise that we can relive it in our imagination: “To produce the material, I undertook the consumption of asparagus over several days. For three days, I ate only asparagus spears, prepared with fat and oil, with a little bread. I drank exclusively beer. The total amount of urine expressed within this period was 5,100 cubic centimeters.”
Isolation of the pure odorous substance failed because of its thermal instability. Some years later, Marceli Nencki (1847–1901), one of the founders of physiological chemistry, took up Hilger’s experiments [3]: “Upon my request, four of the gentlemen working in the laboratory declared themselves willing to replace their meal at noon with only asparagus with butter, in the considerable quantity of seven kilos. Their only drink was tea.”
The urine collected until 8 p.m. was distilled “on a sand bath” and the foul-smelling vapor was conducted directly into an aqueous solution of mercury (II) cyanide. The resulting precipitate reacts with hydrochloric acid to release a gas that, when conducted into an aqueous lead (II) acetate solution, forms the previously known lead salt of methanethiol (3) (earlier known as methyl mercaptan) (see Fig. 3). The odor of methanethiol closely resembled that of asparagus urine. However, Nencki observed that “volatile products that do not possess the odor of pure methyl mercaptan, but also a leek-like smell, escaped at the very beginning of the distillation.”
Methanethiol, which has a very low odor threshold, is added to odorless city gas so that leaking gas is detected immediately.
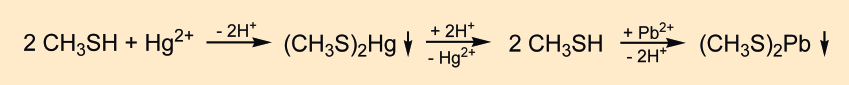 |
|
Figure 3. Necki’s detection of methanethiol. |
Nencki had exhausted the experimental possibilities available in his time and his resignation is discernible in his summary:
“The quantity of lead salt obtained is insufficient for further reactions. However, the facts presented suffice to indicate that methyl mercaptan is most likely the primary source of the singular odor of asparagus urine. It is possible that there are also traces of other sulfur-containing products.”
2. Discovery of the Urine Odor
In the following decades, the puzzle of asparagus urine was considered an amusing curiosity by scientists, and attention turned to other subjects. It took until 1956 for A. C. Allison and K. G. McWhirter to take up the phenomenon of asparagus urine, though not from a chemical point of view. They were interested in the genetics, as not all people can detect the odor of their own asparagus urine [4]. This odor was therefore more than a mere curiosity.
Progress from the chemical point of view was made in the 1980s, when improved analytical methods allowed for gas samples to be collected directly above the surface of a liquid (in the headspace) and injected into a gas chromatograph. The first analyses of this type were carried out by Linus Pauling, who was able to detect 280 compounds in urine and 250 in breath.
After separation, a mass spectrometer could, at least in theory, be used to determine the structure of each individual component [6]. This made it possible to find the structures of the primary volatile components 3–7 of asparagus urine (see Tab. 1) [7,8]. A considerable number of other sulfur-containing compounds were also identified in asparagus urine. However, their low concentration or our low specific sense of smell make them mere supporting players in the overall odor.
|
Table 1. Main components of asparagus urine odor. |
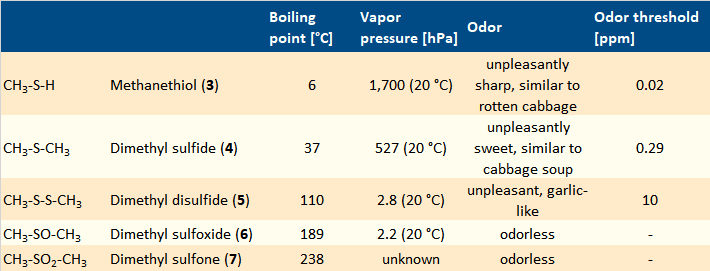 |
Methanethiol (3), dimethyl sulfide (4), and dimethyl disulfide (5) compete for the most unpleasant note in asparagus urine. Their oxidation products, dimethyl sulfoxide (6) and dimethyl sulfone (7), are practically odorless due to their low vapor pressures. In the literature, it is often suggested that dimethyl sulfoxide (6) and dimethyl sulfone (7) contribute a mild, sweet note to the odor of asparagus urine. The fact that these two substances are odorless makes this unlikely.
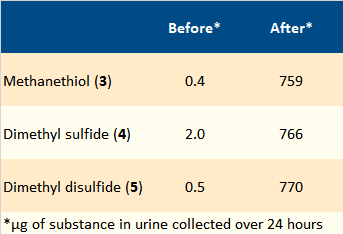
Waring et al. combined water with the sulfur compounds 3–5 to make “synthetic” asparagus urine. It was rated as authentic by test subjects. The same research group confirmed the connection between consumption of asparagus and the urine odor through quantitative measurement of the odorous urine components before and after the ingestion of asparagus. The concentrations of the three odorous components 3–5 did, in fact, rise by a factor of several hundred (see Tab. 2).
|
Table 2. Odorous compounds before and after eating asparagus. |
 |
Our noses are very familiar with the foul-smelling trio 3–5, because they can be smelled in several embarrassing human biogases, including those stemming from flatulence, bad breath, and foot fungus—even when no asparagus has been eaten.
Why does asparagus produce odorous compounds? We will look into this in part 2.
References
[1] Auguste-Arthur Plisson, Étienne-Ossin Henry, Recherches sur les Substances organiques azotées (in French), Ann. Chim. Phys. 1830, 45, 304–321.
[2] Louis-Nicolas Vauquelin, Pierre Jean Robiquet, Essai analytique des asperges (in French), Ann. Chim. 1805, 55, 152–171.
[2a] Louis-Nicolas Vauquelin, Pierre Jean Robiquet, Découverte d’un nouveau principe végétal dans les Asperges (in French), Ann. Chim. 1806, 57, 88–96.
[3] M. Nencki, Ueber das Vorkommen von Methylmercaptan im menschlichen Harn nach Spargelgenuss (in German), Arch. Exp. Pathol. 1891, 28, 206. https://doi.org/10.1007/BF01824333
[4] A. C. Allison, K. G. McWhirter, Two Unifactorial Characters for which Man is Polymorphic, Nature 1956, 178, 748–749. https://doi.org/10.1038/178748c0
[5] L. Pauling et al., Quantitative Analysis of Urine Vapor and Breath by Gas-Liquid Partition Chromatography, Proc. Nat. Acad. Sci. USA 1971, 68, 2374–2376. https://doi.org/10.1073/pnas.68.10.2374
[6] B. Kolb, L. S. Ettre, Static Headspace-Gas Chromatography, John Wiley & Sons, Inc., Hoboken, NJ, USA, 2006. ISBN: 978-0-471-74944-8
[7] R. H. Waring et al., The chemical nature of the urinary odour produced by man after asparagus ingestion, Xenobiotica 1987, 17, 1363–1371. https://doi.org/10.3109/00498258709047166
[8] M. L. Pelchat et al., Excretion and Perception of a Characteristic Odor in Urine after Asparagus Ingestion: a Psychophysical and Genetic Study, Chem. Senses 2011, 36, 9–17. https://doi.org/10.1093/chemse/bjq081
The article has been published in German as:
- Vom Hochgenuss zum Gestank,
Sabine Streller, Klaus Roth,
Chem. unserer Zeit 2018, 52, 112–119.
https://doi.org/10.1002/ciuz.201800853
and was translated by Caroll Pohl-Ferry.
The Smell of Asparagus Urine – Part 1
What substances cause the characteristic smell of asparagus pee?
The Smell of Asparagus Urine – Part 2
Why does asparagus produce odorous compounds?
The Smell of Asparagus Urine – Part 3
How does asparus change when it is cooked?
The Smell of Asparagus Urine – Part 4
Does everybody’s urine smell after eating asparagus?
See similar articles by Klaus Roth published on ChemistryViews.org




