After covering saccharin, the poisonous lead(II) acetate, cyclamate, aspartame, acesulfame-K, thaumatin, sucralose, and dihydrochalcones, we have a look at strategies to find new artificial sweeteners.
20. Intelligent Synthetic Strategies
All the sweeteners presented so far were discovered accidentally. Chemists have subsequently attempted to apply synthetic imagination to their chance discoveries in the hope of increasing the sweetening power further. That actually worked out in the case of acesulfame-K (6) (see Part 7): Starting from the fortuitously encountered sweet molecule 16, removal of one methyl group permitted the development of a new commercially successful sweetener!
In other cases, as with aspartame (see Part 6), thousands of new derivatives were prepared and tasted (!), resulting from variations at every corner and end of the molecule, but all more or less in vain. The effort did indeed produce compounds sweeter than aspartame itself, which was interesting from the standpoint of basic research, but in the development of a commercial sweetener, one dare not lose sight of the profitability of a synthetic approach. Nevertheless, one can only marvel at chemists who refuse to become discouraged in such a quest. This certainly applies to Professor Claude Nofre and Dr. Jean-Marie Tinti from the Claude Bernard University in Lyon, France.
20.1. Superaspartame and Thiosuperaspartame
These chemists set their sights not on fortuitous chance, but attempted rather to draw conclusions from the sweetening powers of their many aspartame variants. They worked from the perspective of spatial demands of presumed “sweet” receptors on our tongues, developing “intelligent” structural proposals. That became the basis for extensive synthesis and tasting, in which “We tasted all of our compounds, literally all of them!” [90].
On the basis of sampling hundreds of aspartame derivatives, they ultimately postulated that, in addition to a binding site for the aspartate fragment and the phenyl ring, there must also exist an additional hydrophobic binding pocket. Model considerations led them accordingly to substitute the free amino group in the aspartate fragment. In 1982, they had the “luck of the industrious”, thereby discovering “superaspartame” 30 (see Fig. 28), with a sweetening power of 14,000.
|
|
|
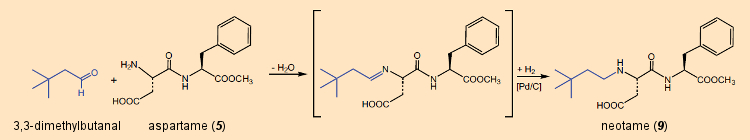
Figure 28. Aspartame becomes even sweeter. |
That proved to be only the beginning, however, since “intelligent search strategies” made it possible to identify other extremely sweet compounds. Thus, in 1985, exchanging the oxygen for sulfur in the urea unit of superaspartame, they achieved a further substantial increase in sweetening power, to an amazing 50,000 in thiosuperaspartame, 31.
20.2. Neotame
Gratifying and admirable as such successes were from the standpoint of basic research, from the perspective of a commercial sweetener other parameters besides mere sweetening power often become decisive: i.e. toxicity, production cost, product quality, and marketing prospects, among others.
It requires little fantasy to realize that makers of the market-leading aspartame-product NutraSweet® – while cooperating with C. Nofre and J.-M. Tinti – were impressed by the sweetening power of superaspartame (30), but at the same time were somewhat reluctant to embrace the scary prospect of a nitrile group with its relationship to hydrogen cyanide. They were also concerned about hard-to-predict issues related to metabolism. Thus, systematic research continued with respect to the hydrophobic substituent, and with constant attention to chain length and branching.
In 1991, persistence on the part of the French researchers paid off: The introduction of a 3,3-dimethylbutyl group led to N-[N-(3,3-dimethylbutyl)-L-α-aspartyl)-L-phenylalanine-1-methyl ester (9) (“neotame”), with a sweetening power of ca. 11,000.
Neotame can be prepared quite simply – and in high yield – from 3,3-dimethylbutanal and aspartame (see Fig. 29). The free amino group of aspartame first initiates a nucleophilic attack on the carbonyl group of 3,3-dimethylbutanal, and subsequent elimination of water leads to a Schiff base, which is catalytically reduced to the alkylated amine. In methanol, this sequence can be carried out at industrial scale in high yield as a “one-pot” reaction [91]. The added synthetic effort is more than repaid by a 50-fold increase in sweetening power.
|
|
|
Figure 29. Synthesis of neotame. |
Comparison with Aspartame
Neotame displays a taste profile similar to that of aspartame, but from a chemical standpoint it is superior to aspartame in two respects:
- Neotame contains no primary amino group, so further formation of a Schiff base is impossible. That, in turn, prevents undesirable reactions of the sweetener with aldehydic flavorings, such as vanillin and cinnamaldehyde.
- As a secondary amine, neotame has no tendency toward formation of dioxopiperazines at higher temperatures (see Part 6, Fig. 18), so that it can be readily utilized in baked goods.
Metabolism
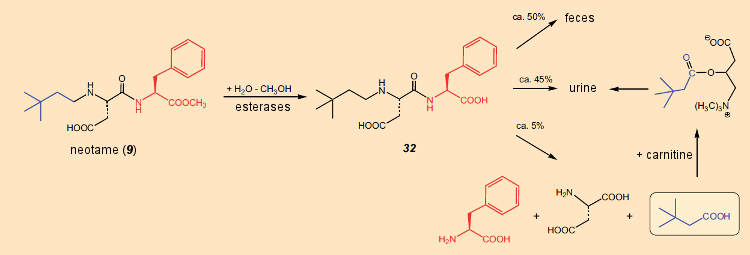
Metabolism of aspartame in the body (see Part 6, Fig. 18) differs fundamentally from that of neotame (see Fig. 30). Aspartame is degraded entirely to methanol, phenylalanine, and aspartic acid. However, whereas esterases do indeed cleave the ester linkage in neotame, forming methanol, most of the free carboxylic acid that results (32) is eliminated unchanged through the urine and feces; only a small amount (< 5 %) is degraded (as in the case of aspartame) to phenylalanine and aspartic acid.
|
|
|
Figure 30. Degradation of neotame in the human body. |
Toxicology
All animal experiments provided good tolerance results for neotame, for both chronic and high single doses. But mice, rats, and dogs were found to reject feed with neotame contents of 5 % or greater. The highest chronic dosage achieved in a one-year-old rat investigation involved 1 g/kg bw/d, corresponding to 70 g of neotame per day. This was the equivalent for an adult of 700 kg of sugar per day. A complete and substantiated presentation of all the available studies was provided by the European Food Safety Authority (EFSA) in 2007 [92].
Use of neotame as a sweetener was authorized in the United States in 2002, and in the EU in 2007. The ADI value (ADI = acceptable daily intake) was established at 2 mg/kg bw/d, representing a sweetening power equivalent to more than 1000 kg of sugar for an adult.
References
[90] B. Bilger, The New Yorker 2006, May 22, 40. Link
[91] C. Nofre, J.-M. Tinti, Food Chem. 2000, 69, 245–257. DOI: 10.1016/S0308-8146(99)00254-X
[92] European Food Safety Authority, EFSA J. 2007, 581, 1–43. DOI: 10.2903/j.efsa.2007.581
The article has been published in German as:
- Die Saccharin-Saga,
Klaus Roth, Erich Lück,
Chem. unserer Zeit 2011, 45, 406–423.
DOI: 10.1002/ciuz.201100574
and
- Kalorienfreie Süße aus Labor und Natur,
Klaus Roth, Erich Lück,
Chem. unserer Zeit 2012, 46, 168–192.
DOI: 10.1002/ciuz.201200587
and was translated by W. E. Russey.
The Saccharin Saga – Part 1
The invention of the first artificial sweetener and a lifetime battle for credit
The Saccharin Saga – Part 2
The early industrial production and organized smuggling of saccharin
The Saccharin Saga – Part 3
The health concerns associated with artificial sweeteners
The Saccharin Saga – Part 4
A glance back to ancient Rome, and the most hair-raising of all sweeteners
The Saccharin Saga – Part 5
What’s in your softdrink? – Introducing cyclamate
The Saccharin Saga – Part 6
Aspartame – a sweet dipeptide ester
The Saccharin Saga – Part 7
Acesulfame-K – another successful sweetening agent
The Saccharin Saga – Part 8
Thaumatin – a sweet protein with a licorice aftertaste
The Saccharin Saga – Part 9
Sucrose or Splenda turned into a low-calorie alternative to sucralose or saccharose
The Saccharin Saga – Part 10
Combining sweet cations and anions, and turning bitter compounds into sweeteners
The Saccharin Saga – Part 11
Intelligent synthetic strategies for low-calorie sweeteners
The Saccharin Saga – Part 12
Stevia plant extracts as low-calorie sweeteners
The Saccharin Saga – Part 13
Finding the best mixture of sweeteners to replicate the taste of real sugar
See all articles by Klaus Roth published in ChemistryViews Magazine

.jpg)